Aldol Condensation in Flavor Chemistry: Mechanisms, Conditions, and Impact on Shelf Life
Aldol condensation is one of a handful reactions the Society of Flavor Chemists requires flavorists to understand and consider when formulating flavors.
1) Chemical Groups Involved and Conditions Required
Key Functional Groups
Aldol condensation is a carbon–carbon bond-forming reaction between carbonyl compounds. The essential groups include:
- Aldehydes (–CH=O)
Example: acetaldehyde, benzaldehyde - Ketones (–CO–)
Example: acetone, diacetyl - α-hydrogen (alpha hydrogen)
A hydrogen attached to the carbon adjacent to the carbonyl group (-C=O) is required for enolization. - Enol / enolate intermediates
Reactive nucleophiles formed under catalytic conditions
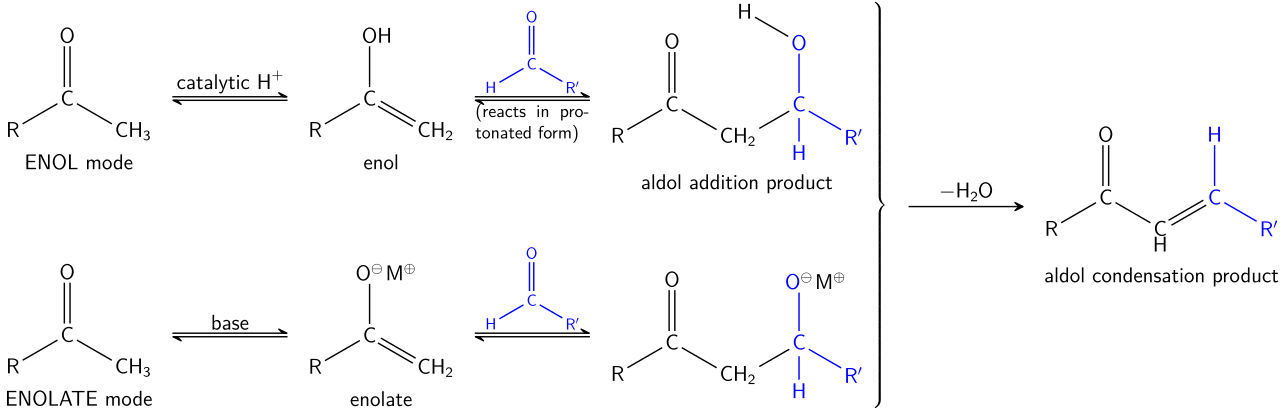
Mechanistic Overview
- Enolate formation (base-catalyzed) or enol formation (acid-catalyzed)
- Nucleophilic attack on another carbonyl compound
- Formation of a β-hydroxy aldehyde or ketone (aldol)
- Dehydration → α,β-unsaturated carbonyl compound (key flavor-active product)
Reaction Conditions
Base-Catalyzed (most relevant in food/flavor systems)
- Mild bases: NaOH, KOH, carbonates
- pH: typically >7
- Temperature: ambient to moderate (20–80°C)
Acid-Catalyzed
- Strong acids (less common in flavor systems)
- pH: <4
- Often slower and less selective
Thermal Conditions
- Heat accelerates:
- Enolate formation
- Dehydration step
Solvent Environment
- Aqueous systems (beverages)
- Alcoholic systems (flavor extracts)
- Low water activity enhances dehydration products
2) Factors Accelerating or Inhibiting Aldol Reactions (and Formulation Considerations)
Factors That Accelerate Aldol Condensation
1. pH (Alkalinity)
- Higher pH → increased enolate formation
- Important in:
- Dairy analogs
- Processed beverages
2. Temperature
- Heat speeds both condensation and dehydration
- Relevant during:
- Pasteurization
- Spray drying
- Flavor compounding
3. Carbonyl Reactivity
- More reactive:
- Small aldehydes (acetaldehyde)
- α-dicarbonyls (diacetyl)
- Less reactive:
- Sterically hindered ketones
4. Concentration of Reactants
- Higher carbonyl concentration → higher reaction probability
5. Low Water Activity
- Promotes dehydration → formation of stable unsaturated compounds
Factors That Inhibit Aldol Condensation
1. Low pH (Acidic Systems)
- Suppresses enolate formation
- Important in:
- Soft drinks
- Fruit beverages
2. Antioxidants
- Indirectly inhibit by stabilizing reactive intermediates
- Examples:
- Ascorbic acid
- Tocopherols
3. Sulfur Compounds
- Can react with carbonyls, reducing availability
- Example:
- Bisulfite addition
4. Steric Hindrance
- Bulky molecules reduce reactivity
5. Water Activity (High Moisture)
- Favors aldol formation but can suppress dehydration step
Formulation Considerations
- Control pH tightly to prevent unwanted reactions
- Use carbonyl scavengers (e.g., sulfites where allowed)
- Optimize thermal processing conditions
- Consider encapsulation to isolate reactive species
- Balance reactive vs stable flavor notes
3) Examples of Aldol Condensation in Flavor Systems
Example 1: Acetaldehyde Self-Condensation
- Reactant: acetaldehyde
- Product:
- 3-hydroxybutanal → crotonaldehyde (after dehydration)
Flavor impact:
- Green, pungent, fatty notes
- Can become harsh at high levels
Example 2: Acetone + Benzaldehyde
- Cross-aldol condensation
- Product: benzalacetone (α,β-unsaturated ketone)
Flavor impact:
- Sweet, floral, balsamic nuances
- Used in perfumery and flavor bases
Example 3: Diacetyl-Based Systems
- Diacetyl participates in condensation reactions under heat
Flavor impact:
- Butter → toasted → caramelized transitions
Example 4: Aldol Reactions in Maillard Systems
- Aldehydes formed during Maillard reactions undergo further aldol condensation
Products:
- Unsaturated aldehydes
- Flavor-active intermediates
Flavor impact:
- Roasted
- Nutty
- Browned notes
Example 5: Citral Degradation Pathways
- Citral (α,β-unsaturated aldehyde) can undergo secondary condensation reactions
Flavor impact:
- Loss of fresh citrus
- Formation of heavier, cooked notes
4) Impact on Flavor Aging and Shelf Life
Positive Effects (Controlled Reactions)
Flavor Development
- Formation of:
- α,β-unsaturated aldehydes and ketones
- Contributes to:
- Complexity
- Warm, aged, rounded profiles
Examples
- Brown flavors (caramel, coffee)
- Aged extracts
Negative Effects (Uncontrolled Reactions)
Flavor Degradation
- Loss of fresh top notes (e.g., citrus, green)
- Formation of:
- Harsh
- Bitter
- Resinous compounds
Off-Flavor Formation
- Over-condensation leads to:
- Polymerized compounds
- “Stale” or “oxidized” perception
Color Changes
- Conjugated systems formed → yellowing or browning
- Important in:
- Clear beverages
- Light-colored products
Stability Concerns
High Risk Systems
- Neutral or alkaline beverages
- Heat-processed flavors
- High aldehyde content systems
Low Risk Systems
- Acidic beverages (pH < 4)
- Encapsulated flavors
- Refrigerated products
Practical Shelf-Life Strategies
1. pH Optimization
- Maintain acidic conditions (if possible)
2. Temperature Control
- Minimize heat exposure post-production
3. Ingredient Selection
- Use less reactive carbonyl compounds
- Replace unstable aldehydes with:
- Acetals (more stable)
4. Encapsulation Technologies
- Spray drying
- Emulsion-based protection
5. Antioxidant Systems
- Reduce secondary reactions that feed aldol pathways
Key Takeaways
- Aldol condensation is a critical reaction in flavor formation and degradation
- It creates important flavor-active unsaturated compounds
- Reaction rate is highly dependent on:
- pH
- Temperature
- Carbonyl availability
- Proper control is essential for:
- Flavor stability
- Shelf life
- Product consistency
Here is a professional, formulation-ready design for a controlled aldol condensation flavor base, targeting caramel, cocoa, and roasted profiles. This approach mimics industrial flavor creation while maintaining reaction control, reproducibility, and shelf stability.
Controlled Aldol Reaction Flavor Base
(Caramel • Cocoa • Roasted System)
1) Design Philosophy
Aldol condensation is used here to deliberately generate:
- α,β-unsaturated aldehydes/ketones → warm, тәт toasted, caramelized notes
- Conjugated systems → depth, color, lingering mouthfeel
- Secondary reaction precursors → cocoa/roasted complexity
We control the system by:
- Selecting specific carbonyl precursors
- Applying mild alkaline catalysis
- Using thermal modulation
- Quenching at optimal flavor peak
2) Core Reaction System (Bench-Scale)
A. Carbonyl Precursor Blend
| Component | Function | Typical Level (%) |
|---|---|---|
| Acetaldehyde (as precursor or generated in situ) | Primary aldol donor | 5–10 |
| Acetone | Ketone partner (cross-aldol) | 10–20 |
| Diacetyl | Butter/caramel precursor + reactive dicarbonyl | 1–3 |
| Furfural | Sweet, almond, bready backbone | 2–5 |
| 5-Methylfurfural | Caramelized sugar depth | 1–3 |
👉 These create:
- Crotonaldehyde derivatives
- Benzalacetone-like structures
- Furan-extended conjugation systems
B. Reaction Medium
- Solvent:
- Water : ethanol (70:30) or propylene glycol system
- Solids (optional):
- Reducing sugars (glucose, fructose) at 2–5% for hybrid Maillard–aldol synergy
C. Catalyst System
- Sodium carbonate or bicarbonate
- Level: 0.2–0.5%
- Target pH: 8.0–9.5
This ensures:
- Controlled enolate formation
- Avoidance of excessive polymerization
D. Thermal Profile
Stage 1: Initiation
- 40–50°C for 30–60 min
- Controlled enolate generation
Stage 2: Condensation
- 70–90°C for 1–3 hours
- Formation of β-hydroxy intermediates
Stage 3: Dehydration / Flavor Development
- 95–110°C for 30–90 min
- Formation of:
- α,β-unsaturated aldehydes
- Light polymeric color bodies
E. Quenching
- Rapid cooling to <40°C
- Adjust pH to 5.0–6.0 using citric acid
This:
- Stops further aldol reactions
- Stabilizes flavor profile
3) Flavor Direction Modulation
A. Caramel Aldol Base
Key Adjustments
- Increase:
- 5-methylfurfural (→ burnt sugar)
- Diacetyl (→ buttery caramel)
- Add:
- Maltol / ethyl maltol (post-reaction)
Reaction Outcome
- Sweet, buttery, brown sugar
- हल toasted sugar crust
Optional Post-Blend (ppm level)
- Ethyl maltol: 50–200 ppm
- Vanillin: 20–80 ppm
B. Cocoa Aldol Base
Key Adjustments
- Increase:
- Furfural + acetone cross-condensation
- Introduce:
- Trace phenylacetaldehyde (floral cocoa lift)
Optional Nitrogen Source (hybridization)
- Add amino acid (e.g., alanine or glycine at 0.5–1%)
→ Enables Maillard + aldol overlap
Reaction Outcome
- Cocoa powder
- Dark chocolate bitterness
- हल nutty dryness
C. Roasted Aldol Base
Key Adjustments
- Increase:
- Acetaldehyde + diacetyl system
- Higher final temperature:
- 105–115°C کوت duration
Add sulfur (optional, very low ppm)
- Thiamine or cysteine (0.05–0.2%)
Reaction Outcome
- Roasted nuts
- Coffee-like top notes
- Toasted grain
4) Example Quantitative Prototype (1000 g Batch)
Reaction Charge
| Ingredient | Amount (g) |
|---|---|
| Water | 500 |
| Ethanol | 200 |
| Acetone | 120 |
| Acetaldehyde (or precursor) | 60 |
| Furfural | 40 |
| 5-Methylfurfural | 20 |
| Diacetyl | 10 |
| Glucose | 30 |
| Sodium carbonate | 5 |
Processing
- Mix all except base → heat to 40°C
- Add base → adjust pH to ~9
- Heat stepwise (as above)
- Monitor aroma every 30 min
- Quench at optimal profile
Post-Reaction Stabilization
| Additive | Level |
|---|---|
| Citric acid | to pH 5.5 |
| Propylene glycol | 5–10% |
| Antioxidant (ascorbic acid) | 0.05% |
5) Key Reaction Products & Flavor Contribution
| Compound Class | Sensory Role |
|---|---|
| α,β-unsaturated aldehydes | Fatty, green, roasted backbone |
| Furan derivatives | Caramel, sweet, baked |
| Conjugated ketones | Warm, lingering depth |
| Light oligomers | Body, color |
6) Shelf Life & Stability Engineering
Risks
- Continued aldol → harshness
- Polymerization → haze, darkening
- Loss of top notes
Control Strategies
1. pH Lock
- Final pH: ≤5.5
2. Oxygen Control
- Nitrogen flushing (industrial scale)
3. Encapsulation (optional)
- Spray dry into:
- Gum arabic / modified starch
4. Carbonyl Management
- Convert reactive aldehydes → acetals (optional for long shelf life)
7) Sensory Tuning Guide
| If you want more… | Do this |
|---|---|
| Sweet caramel | Increase 5-methylfurfural |
| Burnt sugar | Increase temp/time |
| Cocoa bitterness | Increase furfural + amino acid |
| Roasted nuttiness | Add sulfur trace |
| Smoothness | Reduce pH faster after reaction |
8) Industrial Insight
This controlled aldol system is often used as:
- A base note generator
- A reaction flavor intermediate
- A cost-efficient replacement for long-aged extracts
It is frequently combined with:
- Maillard systems
- Lipid oxidation products
- Natural extracts
Final Takeaway
A controlled aldol reaction system allows you to:
- Build caramel, cocoa, and roasted flavors from first principles
- Precisely tune:
- Reaction intensity
- Flavor direction
- Shelf stability
- Replace or enhance traditional reaction flavors with high reproducibility
###