Precipitation in Food & Flavor Systems: Chemistry, Mechanisms, Real-World Examples, and Impact on Shelf Life
Abstract — Precipitation in Food and Flavor Systems
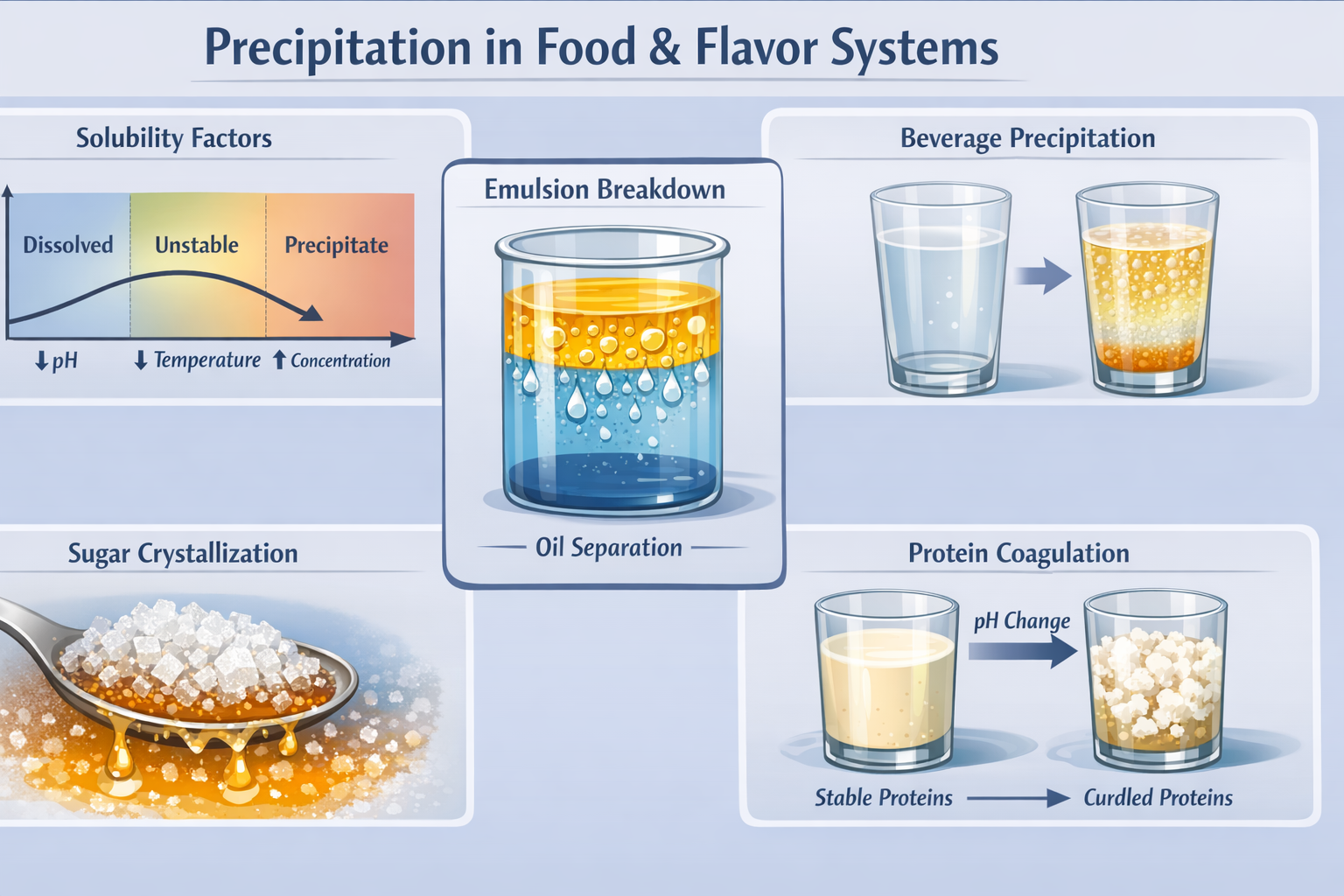
Precipitation is a critical physicochemical process in food and flavor systems in which dissolved or dispersed components lose solubility and separate into a solid or distinct phase. This phenomenon directly impacts product stability, sensory quality, and shelf life, making it an essential topic for flavorists to understand. The process is governed by the behavior of key chemical groups—including carboxylic acids and their salts, proteins, polyphenols, mineral ions, hydrocolloids, sugars, and hydrophobic flavor compounds—each of which responds differently to changes in the surrounding environment.
Precipitation occurs when specific conditions disrupt solubility equilibrium. The most important triggers include pH shifts that alter molecular charge and ionization, temperature changes that reduce solubility, increased concentration leading to supersaturation, variations in solvent polarity during dilution, elevated ionic strength from salts, and interactions between incompatible ingredients such as proteins with polyphenols or calcium with pectin. In many systems, precipitation is not immediate but develops over time through nucleation and crystal growth, often accelerated by storage conditions or physical disturbances.
Real-world examples illustrate the diversity of precipitation mechanisms in flavor applications. These include sugar crystallization in syrups, benzoic or sorbic acid formation in acidic beverages, protein coagulation in dairy and plant-based systems, mineral salt precipitation in fortified drinks, polyphenol haze in tea, and oil separation in citrus flavors upon dilution. Each example demonstrates how formulation variables and environmental conditions influence stability, highlighting the importance of understanding both individual ingredient behavior and system-wide interactions.
Beyond visual defects, precipitation significantly affects flavor aging and shelf life. As components fall out of solution, flavor balance shifts, aroma intensity may decrease, and release becomes uneven. Additionally, precipitation can indirectly accelerate degradation by destabilizing emulsions, exposing reactive compounds, or altering the chemical environment. Over time, these changes lead to non-uniform aging, sensory drift, and reduced consumer acceptance. Therefore, controlling precipitation is not only a matter of appearance but also a key factor in maintaining flavor integrity and product performance throughout shelf life.
In summary, precipitation represents a fundamental stability challenge in flavor formulation. A thorough understanding of the chemical groups involved, the conditions that promote or inhibit the process, and its practical consequences enables flavorists to design more robust systems, anticipate instability risks, and ensure consistent quality from production through consumption.
The following four sections are presented together on one page, making this document somewhat lengthy: 1) Chemical groups involved and conditions for precipitation; 2) Factors affecting precipitation; 3) Examples of precipitation; and 4) How precipitation affects flavors—all of which the Society of Flavor Chemists requires flavorists to understand.
Precipitation in Flavors and Foods
Chemical Groups Involved and Conditions Required
A beginner-friendly guide for flavorist trainees
Precipitation is a process in which a material that was once dissolved in a liquid no longer stays dissolved and comes out as a separate solid phase. In real products, this can look like haze, crystals, floating particles, sediment at the bottom, or a ring on the container wall. In flavor work, precipitation matters because it can make a product look unstable, change flavor release, reduce shelf life, and create customer complaints.
A trainee should remember one core idea:
Precipitation happens when the system can no longer keep a substance dissolved.
That loss of solubility can happen because of pH, temperature, concentration, interactions with other ingredients, solvent changes, salts, minerals, or time.
1) What precipitation looks like in food and flavor systems
Before getting into chemistry, it helps to know what you are looking for.
In foods and flavors, precipitation may appear as:
- fine cloudiness in a beverage
- visible crystals in a syrup
- sediment in the bottom of a bottle
- insoluble particles after dilution
- protein curdling
- botanical extract fallout
- mineral haze
- preservative crystals
- oil-rich material separating and sticking to surfaces
Sometimes precipitation happens quickly. Sometimes the product looks fine at first and fails after days or weeks.
2) Chemical groups involved in precipitation
Different classes of molecules behave differently in water or in flavor systems. A flavorist does not need to memorize advanced physical chemistry first. It is more useful to understand how each group tends to behave and why it may fall out of solution.
A. Carboxylic acids and their salts
What these are
These are compounds that contain a carboxyl group, written as –COOH. In foods and flavors, many important acids belong to this family.
Examples include:
- benzoic acid / sodium benzoate
- sorbic acid / potassium sorbate
- citric acid and citrate salts
- malic acid
- lactic acid and lactates
- fumaric acid
- succinic acid
Why they matter
Carboxylic acids are common in flavor systems because they affect:
- sourness
- preservation
- pH
- buffering
- flavor brightness
- microbial stability
How they are involved in precipitation
These compounds often exist in two forms:
- an ionized form, which is often more water-soluble
- a non-ionized form, which is often less water-soluble
The pH of the system determines which form dominates.
For a trainee, the simple rule is:
When a weak acid becomes less charged, it often becomes less soluble.
Easy example
Sodium benzoate is fairly soluble in water. But in a low-pH beverage, more of it converts into benzoic acid, and benzoic acid is much less soluble. If enough forms, it can precipitate as crystals.
Why this matters in real products
A formula can look stable in the flavor lab but precipitate later when:
- added to an acidic beverage
- stored cold
- combined with other acids or salts
B. Phenolic compounds and polyphenols
What these are
These compounds contain phenolic hydroxyl groups, usually written as Ar–OH, meaning an –OH attached to an aromatic ring.
Examples include compounds from:
- tea
- coffee
- cocoa
- botanicals
- spices
- extracts
- wine materials
Polyphenols include tannins and other plant-derived materials.
Why they matter
Phenolics are important because they contribute:
- bitterness
- astringency
- brown, woody, tea-like, smoky, spicy, or botanical character
- antioxidant behavior
How they are involved in precipitation
Phenolics can react with:
- proteins
- metal ions such as iron or copper
- other polyphenols
When these interactions become strong enough, insoluble complexes form.
Easy way to understand it
Think of polyphenols as molecules that like to grab onto other molecules. If they bind strongly enough to proteins or metals, the new complex may be too heavy or too insoluble to stay dispersed, so it falls out.
Real examples
- tea haze
- wine sediment
- botanical extract clouding
- dark precipitates caused by metal contamination
C. Proteins and peptides
What these are
Proteins are large molecules built from amino acids. They contain many functional groups, especially:
- amino groups, –NH2
- carboxyl groups, –COOH
- side chains that may be acidic, basic, polar, or hydrophobic
Examples in food systems:
- milk proteins such as casein and whey proteins
- plant proteins
- gelatin
- hydrolyzed proteins
- protein-containing extracts
Why they matter
Proteins influence:
- texture
- mouthfeel
- turbidity
- nutrition
- flavor binding
- colloidal stability
How they are involved in precipitation
Proteins stay dispersed partly because of electrical charge and hydration. If the pH moves close to the protein’s isoelectric point, the net charge becomes low. Then protein molecules stop repelling each other and begin to clump together.
Easy way to understand it
When proteins are strongly charged, they stay apart. When they lose that charge, they stick together.
Real examples
- milk curdling in acidic conditions
- protein haze in beverages
- protein fallout in botanical drinks
- precipitates caused by protein-polyphenol interactions
D. Metal ions and mineral salts
What these are
Important mineral ions include:
- calcium, Ca²⁺
- magnesium, Mg²⁺
- iron, Fe²⁺ or Fe³⁺
- copper, Cu²⁺
- potassium, K⁺
- sodium, Na⁺
Why they matter
Minerals are present in:
- processing water
- fortified beverages
- dairy systems
- plant-based beverages
- ingredient impurities
- salts and seasonings
How they are involved in precipitation
Metal ions can react with acids, proteins, polyphenols, phosphates, sulfates, and pectins to form poorly soluble salts or complexes.
Easy example
Calcium can react with citrate or pectin. Even when each ingredient seems fine alone, together they may form insoluble material.
Why trainees should care
Hard water alone can sometimes trigger haze or sediment that never appeared in lab tests using purified water.
E. Phosphates, sulfates, and other inorganic anions
What these are
These are negatively charged groups such as:
- phosphate, PO4³⁻
- sulfate, SO4²⁻
- carbonate, CO3²⁻
- citrate, though organic, also behaves as a multidentate anion in salt formation
Why they matter
They are found in:
- buffers
- mineral systems
- processing aids
- some food ingredients
- fortification systems
How they are involved in precipitation
These anions can combine with multivalent cations like calcium or magnesium to form insoluble salts.
Easy way to understand it
Some positive and negative ions pair up and stay dissolved. Others pair up and create solids.
Examples
- calcium phosphate haze
- calcium citrate precipitate
- mineral instability in fortified drinks
F. Polysaccharides and hydrocolloids
What these are
These are long carbohydrate chains, often used for thickening, stabilizing, suspending, or gelling.
Examples:
- pectin
- carrageenan
- xanthan
- alginate
- gum arabic
- CMC
- starch derivatives
Their important groups are often:
- many hydroxyl groups, –OH
- carboxylate groups in some gums, –COO⁻
- sulfate groups in some hydrocolloids
Why they matter
Hydrocolloids are critical for:
- texture
- suspension
- emulsion stability
- cloud systems
- beverage body
How they are involved in precipitation
They may precipitate or gel when:
- ions neutralize their charge
- calcium cross-links them
- alcohol dehydrates them
- pH moves outside their stable range
- incompatible gums are mixed
- salt concentration becomes too high
Easy example
Pectin can behave well in one beverage but form insoluble complexes when calcium levels are high.
G. Hydrophobic flavor compounds
What these are
These are flavor molecules that do not like water very much. They are often nonpolar or only weakly polar.
Examples:
- terpenes like limonene
- essential oil components
- many aldehydes
- some esters
- some ketones
- oleoresin fractions
Why they matter
These compounds are major contributors to:
- citrus notes
- spice notes
- herbal notes
- floral notes
- top-note aroma
How they are involved in precipitation
Strictly speaking, these may not always form crystalline precipitates, but they often separate because they are insufficiently soluble in water. In practical flavor work, trainees often see this as clouding, oiling out, or insoluble droplets.
Easy way to understand it
A material that prefers oil or alcohol may not stay dissolved when added to water.
Common causes
- dilution of an alcohol-based flavor into water
- not enough emulsifier
- salt effect
- cooling
- wrong carrier system
Real examples
- ring formation in citrus beverages
- cloudy top-note fallout
- insoluble droplets after dilution
H. Sugars and polyols
What these are
Examples:
- sucrose
- glucose
- fructose
- lactose
- sorbitol
- glycerol, though very soluble, still affects solvent balance
Why they matter
Sugars are common in foods and beverages and strongly affect water activity and solubility balance.
How they are involved in precipitation
At high concentrations, sugars themselves can crystallize. They can also change the solvent environment and reduce the solubility of other materials.
Easy example
A syrup may be clear when hot but grow sugar crystals as it cools or ages.
Why trainees should remember this
Even if the flavor ingredient is not the solid that precipitates, sugar may help drive the system toward precipitation by reducing available water.
3) Conditions required for precipitation
Now that the main chemical groups are clear, the next question is: what conditions make precipitation happen?
A. The concentration must approach or exceed solubility
This is the most basic requirement.
Every dissolved substance has a solubility limit in a given system. That limit depends on:
- solvent type
- temperature
- pH
- other dissolved ingredients
When the concentration exceeds what the system can hold, the extra material must come out.
Easy analogy
A parking lot only holds so many cars. Once all spaces are full, extra cars cannot stay there.
In practice
This happens in:
- concentrated syrups
- overloaded flavor bases
- heavily fortified beverages
- cold-stored concentrates
- aged solutions that lose solvent balance
B. The solvent environment must become less favorable
A substance may be soluble in one solvent environment and insoluble in another.
Important factors include:
- water versus alcohol balance
- polarity of the system
- level of humectants or carriers
- presence of oils
- dilution level
Easy example
A flavor dissolved in ethanol may become unstable when diluted into water because the new environment is too polar or otherwise unsuitable.
Why this is common in flavors
Many top-note materials are only partly water-soluble. They require:
- alcohol
- propylene glycol
- emulsification
- encapsulation
- cloud systems
Without the right support, they separate.
C. pH must move the molecule into a less soluble form
This is one of the most important conditions for weak acids, weak bases, proteins, and some hydrocolloids.
How it works
pH changes:
- charge
- ionization state
- intermolecular attraction
- hydration
Easy trainee rule
Charged molecules usually stay in water better than uncharged ones.
So when pH causes a molecule to lose charge, precipitation risk often rises.
Examples
- benzoic acid crystals in low-pH systems
- protein precipitation near the isoelectric point
- hydrocolloid instability outside the proper pH range
D. Temperature must reduce solubility or promote crystallization
Temperature is critical.
Many substances dissolve better at higher temperatures. When the product cools:
- solubility may decrease
- crystals can form
- emulsions may destabilize
- poorly soluble materials may nucleate and grow
Easy example
A syrup looks clear while hot, but crystals appear after cooling.
Real systems affected
- beverage concentrates
- refrigerated drinks
- syrup systems
- essential oil components
- sugar systems
- certain acid salts
E. There must be incompatible ions or ingredients present
A substance may remain dissolved alone but precipitate when another ingredient is present.
Why this happens
The second ingredient may:
- neutralize charge
- form an insoluble salt
- form a large complex
- reduce hydration
- create local concentration shocks
Common incompatible pairs
- calcium + citrate
- calcium + pectin
- protein + polyphenol
- low pH + protein
- metal ions + botanical phenolics
- salt + some hydrocolloids
Easy way to remember
Some ingredients are stable alone but unstable together.
F. The system may require a nucleation trigger
Even if a solution is supersaturated, crystals do not always appear instantly. Often the system needs a starting point, called a nucleation site.
This may be:
- a tiny particle
- a dust speck
- scratches in the container
- an undissolved crystal
- agitation or vibration
Easy way to understand it
Crystals often need a “seed” to start building.
Why this matters
A solution may appear stable until:
- it is shaken
- transported
- cooled
- exposed to tiny particles
G. Time is often required for growth
Precipitation is not always immediate. A system may slowly move from invisible instability to visible failure.
Over time:
- molecules cluster
- nuclei form
- crystals grow
- particles aggregate
- sediment becomes visible
Trainee lesson
A clear product on day one is not automatically a stable product.
4) How the main conditions connect to the chemical groups
For trainees, it helps to connect the groups and conditions together.
Carboxylic acids and salts
Most sensitive to:
- pH
- temperature
- concentration
- common ions
Proteins
Most sensitive to:
- pH
- polyphenols
- salts
- heat
- minerals
Polyphenols
Most sensitive to:
- proteins
- metal ions
- oxidation state
- storage time
Mineral salts
Most sensitive to:
- counterions present
- pH
- concentration
- water hardness
Hydrocolloids
Most sensitive to:
- calcium and other ions
- alcohol
- pH
- incompatible gums
- process order
Hydrophobic flavor compounds
Most sensitive to:
- solvent balance
- dilution
- temperature
- emulsifier level
- salt level
5) Beginner-friendly real examples
Example 1: Preservative crystals in a beverage
A beverage contains sodium benzoate. The system becomes quite acidic. More benzoic acid forms, and benzoic acid is less soluble. Over time, crystals appear.
Example 2: Tea haze
Tea polyphenols interact with proteins or minerals, especially upon cooling. The drink becomes cloudy.
Example 3: Milk curdling
Acid lowers the pH near the isoelectric point of casein. Protein molecules lose repulsion and clump together.
Example 4: Calcium-fortified beverage sediment
Calcium reacts with citrate, phosphate, or pectin and forms an insoluble precipitate.
Example 5: Citrus flavor fallout
A citrus top note dissolved in alcohol is diluted into water. Some oil-rich aroma materials are no longer soluble and separate out.
Example 6: Sugar crystal growth in syrup
A syrup is supersaturated. Cooling and storage allow crystals to nucleate and grow.
6) What flavorist trainees should remember most
When trying to understand precipitation, ask these questions:
What is the material that is falling out?
Is it:
- an acid
- a salt
- a protein
- a mineral complex
- a botanical polyphenol
- a hydrocolloid
- an oil-soluble flavor component
- sugar or another crystal-forming ingredient
Then ask:
What changed?
- pH?
- temperature?
- dilution?
- solvent?
- salt level?
- mineral content?
- storage time?
- ingredient compatibility?
That way of thinking is more useful than trying to memorize isolated facts.
7) Summary
Precipitation in food and flavor systems happens when dissolved materials lose solubility and separate as solids, particles, or visible sediment.
The main chemical groups involved are:
- carboxylic acids and their salts
- phenolics and polyphenols
- proteins and peptides
- mineral ions and inorganic salts
- phosphates, sulfates, and related anions
- polysaccharides and hydrocolloids
- hydrophobic flavor compounds
- sugars and related solids
The main conditions required are:
- concentration reaching or exceeding solubility
- pH shifting a material into a less soluble form
- temperature decreasing solubility
- solvent balance becoming unfavorable
- incompatible ingredients reacting together
- nucleation beginning crystal formation
- time allowing particles or crystals to grow
For a flavorist trainee, the simplest rule is this:
Precipitation is usually a solubility problem caused by chemistry, formulation, or storage conditions.
Once you learn to ask what the material is, what form it is in, and what changed in the system, precipitation becomes much easier to understand and troubleshoot.
Precipitation in Food & Flavor Systems
Factors That Accelerate or Inhibit Precipitation, and What Flavorists Should Consider During Formulation
To understand precipitation, it helps to keep one simple idea in mind:
Precipitation happens when a material can no longer stay comfortably dissolved in the system.
A flavor may look clear and stable on the day it is made, but later become cloudy, form sediment, or grow crystals because something in the formula or storage conditions pushes one ingredient past its solubility limit.
For flavorist trainees, the most useful way to learn this topic is to think in three parts:
- What makes precipitation happen faster
- What slows it down or prevents it
- What to check when designing a formula
1) Factors That Accelerate Precipitation
These are the conditions that make dissolved materials more likely to come out of solution.
A. High Concentration of a Solute
This is one of the most common causes.
When too much of a compound is added to a system, the liquid may no longer be able to hold all of it in dissolved form. At first the formula may still appear clear, especially if mixing is warm or vigorous, but over time the excess material begins to separate.
Beginner way to think about it:
Imagine stirring sugar into tea. At first it all dissolves. But if you keep adding more and more sugar, eventually some stays at the bottom. The liquid has reached its limit.
In food and flavor systems:
This can happen with:
- organic acids
- salts
- preservatives
- sweeteners
- botanical extracts
- essential oil fractions
- emulsifier systems
- flavor actives in concentrates
Why it accelerates precipitation:
Because the system is close to or beyond its solubility capacity. Even a small change in temperature, pH, or dilution can push it over the edge.
What trainees should remember:
A formula that is “just barely soluble” is risky. It may pass initial appearance checks but fail later during storage.
B. Temperature Drop
Many compounds dissolve better at higher temperatures and less well at lower temperatures. So when a product cools down, especially during refrigerated storage or winter shipping, some dissolved material may no longer remain soluble.
Beginner way to think about it:
Hot water can dissolve more sugar than cold water. When the liquid cools, some sugar may come back out as crystals.
In flavors:
Temperature drop can cause:
- clouding in citrus beverages
- crystallization of flavor ingredients
- sediment in concentrates
- instability of certain extracts
- loss of solubility of oils in hydroalcoholic systems
Why it accelerates precipitation:
Cooling lowers molecular motion and often lowers solubility. Molecules that were once dispersed well start gathering together.
Important trainee lesson:
Always test a flavor not only at room temperature, but also under:
- refrigerated conditions
- freeze-thaw conditions if relevant
- elevated storage followed by cooling
A formula stable in the lab may fail in the real world because shipping and storage temperatures vary.
C. pH Change
pH has a major effect on the solubility of many flavor ingredients, especially weak acids, weak bases, proteins, and hydrocolloids.
Beginner way to think about it:
Some molecules dissolve only when they carry a charge. If pH changes, the charge changes. Once the charge is lost, the molecule may no longer like the water phase and can precipitate.
Examples:
- sodium benzoate can convert to benzoic acid at low pH, and benzoic acid is less soluble
- sorbate systems can lose solubility depending on pH
- proteins can clump near their isoelectric point
- pectin behavior changes depending on acidity and mineral content
Why it accelerates precipitation:
Because pH changes the electrical state of molecules. Charged molecules tend to stay dispersed better in water. Neutral molecules often dissolve less well.
Trainee lesson:
You must always ask:
- What is the final product pH?
- What is the pH of the flavor concentrate?
- Will the pH change during shelf life?
A flavor stable in a neutral test solution may precipitate in an acidic beverage.
D. Increased Ionic Strength or Salt Content
Salt can make some dissolved compounds less soluble. This is often called a salting-out effect.
Beginner way to think about it:
Water molecules are busy surrounding the salt ions. That leaves less “help” available to keep other molecules dissolved.
Where it matters:
- savory flavors
- sauces
- brines
- seasoning systems
- high-mineral beverages
- electrolyte drinks
What may happen:
- hydrophobic aroma compounds separate more easily
- emulsions become less stable
- some proteins or hydrocolloids lose dispersion stability
- haze or sediment forms
Why it accelerates precipitation:
Because the dissolved salts change how water behaves. Water becomes less available to solvate some organic compounds.
Trainee lesson:
The same flavor may behave very differently in:
- plain water
- sweetened water
- salted broth
- acidified beverage
- dairy matrix
Never assume solubility in one system means solubility in all systems.
E. Solvent Change
Many flavor ingredients are not very water-soluble, but are soluble in ethanol, propylene glycol, triacetin, or other carriers. If the solvent balance changes, those ingredients may precipitate.
Beginner way to think about it:
A compound may be comfortable in alcohol but uncomfortable in water. If you suddenly add lots of water, it may “fall out.”
Common example:
A flavor concentrate is clear because it is dissolved in alcohol. But once it is added to a water-based beverage, some components precipitate or cause cloudiness.
Why it accelerates precipitation:
Because solubility depends strongly on the polarity of the system. A solvent system that keeps one ingredient dissolved may fail when diluted.
Trainee lesson:
Always distinguish between:
- solubility in the flavor concentrate
- solubility in the final application
These are not the same thing.
F. Presence of Reactive Minerals or Metal Ions
Calcium, magnesium, iron, and copper can interact with acids, polyphenols, proteins, gums, and other ingredients to form insoluble complexes.
Beginner way to think about it:
A dissolved ingredient may be stable alone, but when it meets a mineral, the two form a new solid that does not dissolve.
Examples:
- calcium can precipitate with citrate or pectin
- iron can interact with polyphenols and cause dark precipitates
- metal ions can promote haze in beverages
Why it accelerates precipitation:
Because precipitation is not always caused by “too much of one ingredient.” Sometimes it is caused by a reaction between two ingredients.
Trainee lesson:
Always check the mineral content of the application water and fortified systems. Hard water can create stability problems that were not seen in deionized lab water.
G. Ingredient Interactions
This is a very important practical factor. Even when each ingredient is soluble by itself, two or more ingredients together may become unstable.
Common interactions:
- protein + polyphenol
- calcium + pectin
- acid + protein
- salts + gums
- alcohol dilution + essential oils
- preservatives + pH changes
Beginner way to think about it:
Each ingredient is not acting alone. A formula is like a crowded room. Some ingredients get along, and some do not.
Why it accelerates precipitation:
Because interactions can create:
- insoluble complexes
- charge neutralization
- dehydration of biopolymers
- phase separation
- crystal nucleation sites
Trainee lesson:
Compatibility is just as important as individual solubility.
H. Time
Some systems do not precipitate right away. They may look fine for days or weeks and then slowly develop haze or sediment.
Beginner way to think about it:
At first, tiny particles may be too small to see. Over time, they grow and become visible.
Why it accelerates visible failure:
Time allows:
- nucleation
- crystal growth
- particle aggregation
- slow reactions
- changes in solvent distribution
Trainee lesson:
A one-day clear result is not proof of stability. Real stability needs storage testing.
I. Agitation, Shock, or Seed Crystals
Physical disturbance can sometimes promote precipitation.
Beginner way to think about it:
If a system is close to unstable, shaking, vibration, or the presence of a small crystal can trigger more crystal growth.
Examples:
- shipping vibration
- repeated pumping
- container wall defects
- undissolved particles acting as starting points
Why it accelerates precipitation:
Because particles need a starting point to build on. Once crystal formation begins, more material may deposit there.
Trainee lesson:
Good filtration and complete dissolution are important. Tiny undissolved particles can act as nuclei.
2) Factors That Inhibit or Slow Precipitation
Now let us look at what helps keep a system stable.
A. Proper Solvent Selection
Choosing the right solvent or carrier is one of the strongest tools a flavorist has.
What it means:
A solvent system can be designed so that difficult ingredients stay dissolved more effectively.
Common carriers:
- ethanol
- propylene glycol
- triacetin
- glycerin
- water
- oil, depending on the flavor type
Why it inhibits precipitation:
Because the right solvent provides a better environment for the target molecules.
Trainee lesson:
Do not choose a carrier only because it is familiar. Choose it because it matches the chemistry of the ingredients and the final application.
B. pH Control
Keeping the system within the right pH range can prevent ingredients from converting into poorly soluble forms.
Why it helps:
Because many materials are most soluble only within a certain pH window.
Example:
A preservative salt may remain soluble in one pH range but not in another.
Trainee lesson:
Know the pH-sensitive ingredients in your flavor. Then ask whether the final product pH keeps them in their soluble form.
C. Lowering Concentration
Reducing the level of a difficult ingredient is often the simplest solution.
Why it helps:
Because a system farther below the solubility limit is more stable.
Trainee lesson:
Sometimes the best formula is not the strongest one. Overloading an impact material can damage stability more than it improves flavor.
D. Use of Emulsifiers or Stabilizers
For hydrophobic flavor compounds, emulsifiers help disperse oils into small droplets rather than trying to dissolve them molecularly in water.
Why it helps:
Because some ingredients are better handled by emulsification than by true dissolution.
Stabilizers may include:
- gum arabic
- modified starch
- weighting agents where allowed and appropriate
- selected hydrocolloids
- surfactants depending on system and regulations
Trainee lesson:
If a compound does not truly want to dissolve in water, forcing it into a clear aqueous system may fail. A stable emulsion may be the better design.
E. Chelating Agents
Chelators bind metal ions so they cannot react as easily with other ingredients.
Example:
EDTA is commonly used in some systems to tie up trace metals.
Why it helps:
Because it reduces metal-driven precipitation and sometimes also slows oxidation-related instability.
Trainee lesson:
If metal ions are causing trouble, removing or tying up the metals may solve the problem more effectively than adjusting the flavor ingredients themselves.
F. Protective Colloids and Hydrocolloids
Some stabilizers help keep small particles apart and suspended.
Why it helps:
They can reduce aggregation and delay sediment formation.
Trainee lesson:
These materials do not always “solve solubility,” but they may improve physical stability enough for a practical product.
G. Better Ingredient Order and Processing Conditions
Sometimes a formula fails not because the ingredients are wrong, but because they were added in the wrong way.
Examples:
- adding acid too early
- adding calcium before full hydration of a gum
- diluting alcohol solutions too quickly
- poor mixing
- insufficient dissolution time
Why it helps:
Good process control avoids local high concentrations and sudden incompatibility.
Trainee lesson:
How a formula is made can be as important as what is in it.
H. Temperature Management
Keeping processing and storage within a safe range can reduce precipitation risk.
Why it helps:
Because sudden cooling or repeated temperature cycling can push a marginal system into instability.
Trainee lesson:
Know both the warm-process behavior and the cold-storage behavior.
3) Formulation Considerations for Flavorists
This is the most practical section. When building a flavor or evaluating a stability problem, these are the questions a flavorist should ask.
A. What Is the Final Application, Not Just the Flavor Concentrate?
A flavor may be stable in its own bottle but unstable in the finished food or beverage.
Important questions:
- Is the final system water-based, oil-based, or emulsion-based?
- Is it acidic?
- Is it salty?
- Is it high in sugar?
- Does it contain proteins, minerals, or fibers?
- Will it be diluted before use?
Trainee lesson:
Always formulate for the real use condition, not just the concentrate.
B. What Is the Expected Shelf Life?
A flavor stable for one week may not be stable for six months.
Questions to ask:
- How long must it remain clear or uniform?
- At what temperatures?
- In what packaging?
- Will it face shipping stress?
Trainee lesson:
Shelf life is not just about flavor strength. It is also about physical appearance and phase stability.
C. Is Clarity Required?
This is critical in beverage work.
Why it matters:
Some systems can tolerate slight cloud or sediment, while others cannot.
A cloudy botanical drink may be acceptable in one category, but a clear beverage cannot tolerate even slight haze.
Trainee lesson:
Know the visual requirement. That determines how much precipitation risk is acceptable.
D. Are There pH-Sensitive Ingredients?
A trainee should always identify these early.
Ingredients often worth checking:
- benzoates
- sorbates
- organic acids
- proteins
- botanical extracts
- certain colorants
- minerals
- hydrocolloids
Trainee lesson:
A formula with many pH-sensitive ingredients needs tighter control and more testing.
E. Are There Mineral Interactions?
Especially important in:
- fortified beverages
- dairy systems
- plant-based beverages
- hard-water applications
- savory systems
Trainee lesson:
Minerals can create problems that do not show up in simple bench tests.
F. Is an Emulsion Better Than a Clear Solution?
This is a smart design question.
Why it matters:
Some aroma compounds are naturally hydrophobic. Trying to force them into a clear aqueous system may be unrealistic.
Trainee lesson:
Choose the right physical format:
- true solution
- emulsion
- encapsulated system
- dry flavor delivery system
Do not fight the chemistry.
G. What Happens During Dilution?
Many precipitation problems appear only when a concentrate is diluted into the food or beverage.
Trainee lesson:
Always do dilution testing at realistic use levels. A clear concentrate can still fail instantly in the final matrix.
H. What Is the Order of Addition?
Order matters because local concentrations can temporarily become extreme.
Example:
If acid is added directly onto a protein-rich or gum-containing system, it may cause local precipitation even if the final average pH would have been acceptable.
Trainee lesson:
Avoid sudden local incompatibility. Premix and add gradually when needed.
I. What Type of Water Is Used?
Water is often underestimated.
Why it matters:
Tap water, process water, and deionized water can behave very differently because of mineral content.
Trainee lesson:
Test with the water the factory will actually use.
J. Have You Tested Under Real Stress Conditions?
A proper formulation review should include:
- room temperature
- refrigeration if relevant
- heat exposure if relevant
- freeze-thaw if relevant
- light exposure if relevant
- shipping agitation if relevant
Trainee lesson:
The formula does not just live in the lab. It lives in trucks, warehouses, factories, and consumer kitchens.
4) A Simple Practical Way for Trainees to Think About Precipitation
When you see haze, crystals, or sediment, ask these questions in order:
First: Is something overloaded?
Maybe the ingredient level is simply too high.
Second: Did pH change the ingredient into a less soluble form?
Especially important for weak acids, salts, proteins, and some extracts.
Third: Did temperature change?
Cooling often reveals hidden instability.
Fourth: Did another ingredient react with it?
Think calcium, metals, proteins, tannins, gums, acids.
Fifth: Did dilution or solvent change cause it?
Very common in beverage flavors.
Sixth: Did time allow crystals or particles to grow?
A slow-forming failure may not appear right away.
This approach helps trainees troubleshoot more systematically.
5) Summary
Factors that accelerate precipitation include:
- high concentration
- low temperature
- unfavorable pH
- high salt or ionic strength
- solvent changes
- mineral interactions
- ingredient incompatibility
- time
- physical shock or crystal seeding
Factors that inhibit precipitation include:
- proper solvent choice
- correct pH control
- lower solute levels
- emulsifiers and stabilizers
- chelating agents
- good process order
- suitable temperature control
- realistic stability testing
The biggest formulation lesson for flavorists is this:
Precipitation is rarely random.
It usually happens because the chemistry, the process, or the storage conditions no longer support solubility.
A good flavorist learns to ask:
- What is dissolved?
- In what kind of system?
- Under what pH, temperature, and dilution?
- In the presence of what other ingredients?
- For how long?
That is how precipitation problems are predicted and prevented.
Examples of Precipitation in Food and Flavor Systems
A beginner-friendly guide for flavorist trainees
To understand precipitation, it helps to move from theory to real examples.
A flavor trainee usually understands the subject much faster once they can see what is precipitating, why it precipitates, what it looks like, and why it matters in real products.
The easiest way to think about precipitation is this:
Something was staying dissolved, dispersed, or suspended before, but the system changed, and now it cannot stay there anymore.
That change may produce:
- crystals
- haze
- sediment
- clumps
- fallout
- cloudy rings
- particles at the bottom of the container
Below are the most important practical examples.
1) Sugar crystallization in syrups and concentrates
What happens
A syrup may look clear when it is freshly made, but later sugar crystals begin to form. These crystals may appear:
- at the bottom of the bottle
- on the side wall
- around the cap
- throughout the syrup
Why it happens
Sugar has a solubility limit. If too much sugar is dissolved, especially at high temperature, the solution may become supersaturated. When it cools or sits over time, some of the sugar comes back out as crystals.
Easy way to understand it
Hot water can hold more sugar than cold water. So a syrup that is stable while warm may become unstable when it cools.
What trainees should notice
This is one of the simplest examples of precipitation because the solid that forms is easy to identify: sugar itself.
Why it matters in flavor work
Even if the flavor is not the thing precipitating, sugar crystallization can:
- ruin texture
- change appearance
- trap or exclude flavor compounds
- affect pouring and processing
- shorten shelf-life acceptability
2) Benzoic acid crystals in acidic beverages
What happens
A beverage containing sodium benzoate may develop visible crystals or sediment after storage.
Why it happens
Sodium benzoate is the salt form and is relatively water-soluble. In an acidic beverage, part of it converts to benzoic acid, and benzoic acid is much less soluble. If enough benzoic acid forms, it can precipitate.
Easy way to understand it
The ingredient changes from a form that likes water more to a form that likes water less.
What trainees should notice
The same ingredient can behave differently depending on pH. In one system it stays dissolved, and in another it may crystallize.
Why it matters in flavor work
Flavorists often work with acidified beverages, syrups, and concentrates. If they do not consider pH-sensitive solubility, preservative crystals may appear during storage.
3) Sorbic acid precipitation from sorbate systems
What happens
A system containing potassium sorbate may later show crystals or haze.
Why it happens
Like benzoate systems, sorbate systems depend strongly on pH. The more acidic the system becomes, the more the preservative shifts toward sorbic acid, which is less soluble than its salt form.
Easy way to understand it
The acid form is less comfortable in water than the salt form.
Why it matters
This teaches trainees an important lesson:
A formula ingredient is not always stable just because it dissolves during mixing.
Its chemical form may change after formulation.
4) Protein precipitation in acidified dairy drinks
What happens
A milk-based or protein-containing beverage may curdle, form fine particles, or develop sediment.
Why it happens
Proteins stay dispersed because they carry charge and are hydrated by water. When the pH approaches the protein’s isoelectric point, the protein loses much of its charge. Then the protein molecules stop repelling each other and begin to clump.
Easy way to understand it
When proteins are charged, they stay apart. When they lose charge, they stick together.
Common example
Adding acid to milk can cause casein to coagulate or precipitate.
Why it matters in flavor work
Many flavored beverages now contain:
- dairy proteins
- plant proteins
- nutritional proteins
- protein hydrolysates
Flavorists must understand that acidity, mineral content, and added botanicals can all affect protein stability.
5) Tea haze from polyphenol interactions
What happens
A tea beverage may be clear while hot, but after cooling it becomes cloudy or forms sediment.
Why it happens
Tea contains polyphenols, which can interact with:
- proteins
- minerals
- caffeine-related complexes
- other tea solids
Cooling can reduce solubility or strengthen interactions enough to produce visible haze.
Easy way to understand it
The tea compounds are no longer staying comfortably dissolved when the system gets colder or when they bind with other compounds.
Why it matters
This is a very important real-world example because haze is not always a sign of contamination. Sometimes it is simply precipitation caused by normal tea chemistry.
Trainee lesson
A product may pass room-temperature testing and still fail under refrigeration.
6) Wine sediment and tannin precipitation
What happens
Wine may develop sediment over time. This sediment may include:
- tartrate crystals
- tannin-related precipitates
- pigment-polyphenol complexes
Why it happens
Wine is chemically complex. Acids, minerals, polyphenols, alcohol, and time all interact. Some compounds become less soluble and slowly fall out.
Easy way to understand it
The system ages, molecules find partners to bind with, and the new materials do not stay dissolved.
Why it matters for flavor trainees
Even if you do not work in wine, this is a classic example showing that precipitation can be:
- slow
- natural
- chemistry-driven
- affected by temperature and storage time
7) Calcium citrate precipitation in fortified beverages
What happens
A calcium-fortified beverage develops cloudiness, fine sediment, or a heavier bottom deposit.
Why it happens
Calcium can react with citrate to form poorly soluble calcium citrate under certain conditions. The risk depends on:
- concentration
- pH
- temperature
- total ionic environment
- presence of other solids
Easy way to understand it
Two ingredients that look fine separately can combine to make a new solid.
Why it matters
Fortified beverages are common, and calcium is one of the most common causes of mineral precipitation problems.
Trainee lesson
Always think about ingredient interactions, not just the solubility of each ingredient by itself.
8) Calcium-pectin precipitation or gel-like fallout
What happens
A fruit beverage or system containing pectin may become unstable, forming clumps, stringy particles, gel pieces, or sediment.
Why it happens
Pectin can interact strongly with calcium. Calcium can link pectin chains together, and this may lead to gel formation or precipitation depending on the system.
Easy way to understand it
Calcium acts like a bridge that ties pectin molecules together.
Why it matters
This is especially relevant in:
- fruit preparations
- juice systems
- cloudy beverages
- fiber-containing systems
Trainee lesson
Hydrocolloids are not just thickeners. They are chemically active materials whose stability depends on pH, salts, and processing conditions.
9) Mineral haze from hard water
What happens
A beverage or flavor application looks fine in lab water but becomes cloudy or forms sediment in production.
Why it happens
Production water may contain calcium, magnesium, iron, or other minerals. These can react with acids, polyphenols, hydrocolloids, or phosphates and form insoluble materials.
Easy way to understand it
The water itself brought in extra reactive ingredients.
Why it matters
This is a very practical lesson for trainees: water is not always just “water.”
Trainee lesson
Always test with the real process water when possible.
10) Citrus oil fallout after dilution
What happens
A citrus flavor concentrate may be clear in the bottle, but when added to water it becomes cloudy, forms droplets, or leaves an oil ring.
Why it happens
Many citrus aroma compounds are hydrophobic. They may dissolve in alcohol or other carriers inside the concentrate, but after dilution into water they are no longer soluble enough.
Easy way to understand it
The flavor materials liked the original solvent system, but they do not like the final one.
Why it matters
This is one of the most important examples in flavor work because many top-note materials are only partly water-soluble.
Trainee lesson
A flavor can be stable in the concentrate and unstable in the application. Those are two different questions.
11) Oiling out in beverage flavors
What happens
A beverage shows small floating droplets, an oily surface layer, or rings on the container wall.
Why it happens
This may not always be a crystal-type precipitation, but in practical flavor work it is often treated as part of the same family of instability problems. Hydrophobic flavor compounds are insufficiently dissolved or insufficiently emulsified and separate from the water phase.
Easy way to understand it
The oil-like flavor compounds are escaping from a system that cannot hold them.
Why it matters
It affects:
- appearance
- flavor uniformity
- aroma release
- consumer acceptance
Trainee lesson
Sometimes the right solution is not to force dissolution, but to change to an emulsion or cloud system.
12) Flavor extract fallout in botanical systems
What happens
A botanical extract may look fine initially but later form sediment, haze, or dark particles.
Why it happens
Botanical extracts often contain many different classes of materials:
- polyphenols
- waxes
- resins
- pigments
- essential oil fractions
- carbohydrates
- proteins
- minerals
When the system changes through dilution, cooling, pH shift, or time, some of these materials become insoluble.
Easy way to understand it
Botanical extracts are chemically crowded systems. Some components remain happy in solution, and others do not.
Why it matters
Natural flavor systems often have higher precipitation risk than very simple synthetic systems because they contain more chemically diverse components.
13) Tartrate crystal formation in grape systems
What happens
Grape juice concentrates or wines may form crystals, especially during cold storage.
Why it happens
Potassium bitartrate and related salts can become less soluble under certain conditions, especially at lower temperatures.
Easy way to understand it
The acid-mineral balance changes, and the system cannot keep the salt dissolved.
Why it matters
This is a classic example of salt precipitation driven by temperature and composition.
14) Lactose crystallization in dairy-related systems
What happens
Dairy concentrates, confections, or dairy-based ingredients may develop gritty texture because lactose crystals form.
Why it happens
Lactose is less soluble than some other sugars, so when its concentration becomes high enough, especially during cooling or drying, crystals can form.
Easy way to understand it
Too much lactose is present for the available water and temperature conditions.
Why it matters in flavor work
Texture changes caused by sugar or dairy-solid precipitation can change flavor perception and mouthfeel.
15) Spice oleoresin fallout
What happens
A spice flavor or seasoning liquid develops sediment or visible dark resinous material.
Why it happens
Oleoresins can contain:
- resin fractions
- heavy oil fractions
- waxes
- pigments
- poorly water-soluble compounds
Changes in solvent balance, temperature, or dilution can cause these heavier fractions to precipitate.
Easy way to understand it
Not every part of a spice extract behaves the same way. Some parts dissolve well, and some parts drop out.
Why it matters
This is common in natural savory and spice systems.
16) Cloud failure in emulsified flavors
What happens
A beverage cloud or emulsion becomes unstable. Over time, particles may clump, cream, or sediment.
Why it happens
In some systems, destabilization of the emulsion lets droplets merge or interact with other ingredients. The result may look like precipitation even though the original issue was colloidal instability.
Easy way to understand it
The system that was keeping oil droplets tiny and separated stopped doing its job.
Why it matters
Flavor trainees should learn that not every visible solid came from true crystallization. Sometimes a failed emulsion creates a precipitation-like appearance.
17) Cocoa or chocolate sediment in beverages
What happens
Chocolate milk or cocoa beverages may form bottom sediment.
Why it happens
This can involve several processes:
- insoluble cocoa solids settling
- protein interactions
- mineral effects
- sugar crystallization in some systems
- poor suspension stability
Easy way to understand it
Some particles were never truly dissolved, only suspended. Once suspension stability weakens, they settle out.
Why it matters
This teaches an important distinction:
not all sediment is true precipitation from a dissolved state.
But in product troubleshooting, the appearance may be similar, so trainees need to learn to tell the difference.
18) Acid-induced fallout in plant-protein beverages
What happens
A plant-protein beverage develops flocculation, graininess, or sediment after flavoring or acidification.
Why it happens
Plant proteins can be sensitive to:
- pH
- salts
- polyphenols
- processing conditions
- heat history
When they lose stability, they aggregate and fall out.
Easy way to understand it
The protein was barely staying dispersed, and the formula change pushed it past its limit.
Why it matters
Modern beverage development often involves highly interactive systems, so flavorists need to understand how acidulants, botanicals, and mineral additions affect proteins.
19) Preservative or acid crystal growth during cold storage
What happens
A product stored cold develops tiny sparkling crystals that were not visible before.
Why it happens
Cooling often lowers solubility. Materials that were just barely dissolved at room temperature may begin to crystallize in the refrigerator.
Easy way to understand it
Cold storage reveals weak points in solubility.
Why it matters
A product can pass ordinary bench testing and still fail once consumers refrigerate it.
20) Slow precipitation during shelf life
What happens
A product looks stable for weeks, then gradually becomes hazy or develops a small amount of sediment.
Why it happens
Some systems do not fail immediately. Instead:
- tiny nuclei form slowly
- crystals grow gradually
- complexes build over time
- small particles aggregate into visible particles
Easy way to understand it
At first the problem is too small to see. Later it grows large enough to notice.
Why it matters
This teaches trainees why real-time and accelerated stability testing are important.
21) A simple way for trainees to classify examples
When you see a precipitation-like problem, try placing it into one of these buckets:
A. True crystal precipitation
Examples:
- sugar crystals
- benzoic acid crystals
- sorbic acid crystals
- tartrate crystals
- lactose crystals
These involve a dissolved material forming an organized solid.
B. Insoluble salt or complex formation
Examples:
- calcium citrate
- calcium phosphate
- protein-polyphenol complexes
- metal-phenolic precipitates
These involve chemical interaction between ingredients.
C. Aggregation or coagulation
Examples:
- milk curdling
- plant-protein fallout
- hydrocolloid clumping
These involve molecules sticking together and forming larger particles.
D. Hydrophobic separation or fallout
Examples:
- citrus oil fallout
- oil rings
- oleoresin separation
These come from poor solubility or poor emulsification.
E. Suspension failure that looks like precipitation
Examples:
- cocoa settling
- cloud failure
- pulp settling
These are not always true precipitation from a dissolved state, but they can look very similar.
This classification helps trainees think more clearly during troubleshooting.
22) What flavorist trainees should learn from these examples
The biggest lesson is that precipitation is not one single event. It is a family of instability problems that can happen for different reasons.
From the examples above, trainees should remember:
- Some precipitation is caused by too much material dissolved
- Some is caused by pH change
- Some is caused by cooling
- Some is caused by ingredient interaction
- Some is caused by dilution into the wrong solvent environment
- Some develops only with time
- Some visible sediment is not true precipitation at all, but poor suspension or emulsion stability
That is why good troubleshooting always begins with a question:
What exactly is the solid or haze made of?
Once you know that, the cause becomes much easier to understand.
23) Summary
Examples of precipitation in foods and flavors include:
- sugar crystals in syrup
- benzoic acid crystals in acidic beverages
- sorbic acid precipitation
- protein precipitation in dairy or plant systems
- tea haze from polyphenols
- wine sediment
- calcium citrate fallout in fortified drinks
- calcium-pectin instability
- hard-water mineral haze
- citrus oil fallout after dilution
- botanical extract sediment
- tartrate crystals in grape systems
- lactose crystallization
- spice oleoresin fallout
- cloud and emulsion failure
- slow shelf-life precipitation
For a trainee, the simplest way to understand all of these is this:
A system changed, and the ingredient could no longer stay dissolved, dispersed, or suspended.
That change may involve:
- pH
- temperature
- concentration
- salts
- minerals
- dilution
- solvent balance
- time
- ingredient compatibility
Once you learn to connect the example to the cause, precipitation becomes much easier to predict and prevent.
How Precipitation Affects Flavor Aging and Shelf Life
A beginner-friendly guide for flavorist trainees
Precipitation is not only an appearance problem. It can also change how a flavor smells, tastes, releases aroma, and survives over time. That is why flavorists care about it so much.
A trainee should remember this simple idea:
When part of a flavor system precipitates, the product is no longer the same system it was at the beginning.
Once material falls out of solution, several things can change:
- flavor balance
- aroma strength
- mouthfeel
- appearance
- chemical stability
- shelf-life acceptance
So precipitation is important not just because consumers can see it, but because it can change the entire performance of the product during storage.
1) Precipitation changes flavor balance
This is one of the most direct effects.
When a material precipitates, it is no longer evenly distributed in the product. That means the flavor system becomes unbalanced.
Easy way to understand it
Imagine making a soup and having some of the seasoning settle at the bottom. The liquid at the top no longer tastes the same as the whole soup. The same idea applies in flavor systems.
In practice
If certain flavor-related materials precipitate, the product may lose:
- brightness
- top note
- sourness
- bitterness
- body
- botanical character
- preservative performance
At the same time, other notes may seem stronger simply because the original balance has changed.
Example
If a citrus beverage loses some hydrophobic top-note material by fallout or oiling out, the drink may smell:
- flatter
- less juicy
- less fresh
- less natural
Even if the rest of the formula is still present, the character is no longer the same.
2) Precipitation can reduce flavor intensity
When a compound is dissolved properly, it is available to move into the headspace and be smelled or tasted in a predictable way. Once it precipitates, clumps, crystallizes, or separates, its availability changes.
Easy way to understand it
A flavor material that is trapped in a crystal or sediment is not as free to contribute to aroma as it was when it was well dissolved or well dispersed.
What this can cause
- weaker aroma
- slower flavor release
- non-uniform flavor from sip to sip
- reduced immediate impact
- lower perceived freshness
Example
If a botanical extract throws sediment, some of the flavor-active components may be tied up in that sediment. The beverage may then taste weaker or duller than it did when freshly made.
3) Precipitation can make flavor release less predictable
Flavor release depends a lot on where the molecules are and what phase they are in.
If the system changes from:
- fully dissolved
- finely emulsified
- evenly suspended
to:
- partially precipitated
- partially settled
- partly stuck on the package wall
then flavor release becomes less controlled.
Easy way to understand it
Instead of one stable flavor system, you now have several mini-systems:
- some material still in the liquid
- some on the bottom
- some on the top
- some attached to the bottle
Each part behaves differently.
Why that matters
A consumer may notice:
- the first serving tastes different from the last
- one bottle tastes stronger than another
- shaking changes the flavor dramatically
- aroma fades faster than expected
This is a shelf-life problem because the product no longer performs consistently.
4) Precipitation can speed up flavor aging indirectly
Precipitation does not always destroy flavor by itself, but it often creates conditions that make aging worse.
When ingredients fall out, separate, or clump, the remaining system becomes less stable. That can lead to faster oxidation, interaction, or loss of volatile compounds.
Easy way to understand it
A stable system protects flavor materials better. An unstable system exposes them.
How this happens
Precipitation may:
- separate protective components from the flavor
- expose oils to oxygen
- remove buffering effects
- disturb emulsion stability
- concentrate reactive substances in one place
- increase contact between certain ingredients that should not be interacting
Example
If an emulsion begins to fail and oil-rich droplets separate, those droplets may become more vulnerable to oxidation. That can lead to:
- stale notes
- terpene oxidation off-notes
- harshness
- loss of freshness
So precipitation can be both a direct problem and the beginning of other shelf-life failures.
5) Precipitation can lead to non-uniform aging
This is a very important concept for trainees.
When the formula is no longer evenly mixed, not all parts of the product age at the same rate.
Easy way to understand it
Think of a beverage bottle with sediment at the bottom. The bottom layer is chemically different from the top layer. That means the bottom and top may age differently.
What may happen
- the top becomes weaker in flavor because some materials have settled out
- the bottom becomes overly concentrated in solids
- reactive components may collect together in one phase
- the aroma profile changes unevenly through storage
Consumer effect
One bottle may taste normal at first and then suddenly seem too strong, too weak, gritty, or chemically unbalanced after shaking or near the end of the container.
This reduces real shelf-life quality even if the product is still technically safe.
6) Precipitation can change the flavor profile over time
Aging is not only about losing strength. It is also about changing character.
When some compounds precipitate and others stay dissolved, the flavor profile shifts.
Easy way to understand it
The “recipe” inside the product changes during storage.
What this means in sensory terms
The product may become:
- less bright
- less fruity
- less floral
- less fresh
- more flat
- more heavy
- more bitter
- more astringent
- more harsh
- less balanced
Example
If delicate top-note materials are lost through separation while heavier base notes remain, the flavor may become dull and bottom-heavy.
This is one of the reasons a product can still “have flavor” but no longer taste good.
7) Precipitation can hurt appearance, and appearance affects shelf-life acceptance
Shelf life is not only chemistry. It is also whether the product still looks acceptable to the customer.
A beverage that develops:
- haze
- crystals
- sediment
- floating particles
- ring formation
may be rejected by consumers even if the flavor is still mostly present.
Easy way to understand it
A product’s useful shelf life ends when people no longer want to use it, not only when a lab says it changed.
Why this matters for flavorists
Flavorists must think about physical stability because it affects:
- product acceptance
- brand trust
- complaints
- returns
- the impression of freshness and quality
A product that looks unstable will often be judged as old, spoiled, or poor quality.
8) Precipitation can interfere with preservatives and acid systems
Sometimes what precipitates is not only flavor material. It may be:
- a preservative
- an acid salt
- a mineral
- a buffering component
When that happens, the shelf life of the product may change in more serious ways.
Easy way to understand it
If a functional ingredient falls out, it may no longer do its job properly.
Example
If benzoic acid or sorbic acid precipitates, the preservative may no longer be distributed in the liquid the way it should be. This can reduce preservative effectiveness or at least make system performance less predictable.
Why trainees should care
A precipitation problem can sometimes become:
- an appearance issue
- a flavor issue
- a shelf-life issue
- and a microbiological concern
all at once.
9) Precipitation can increase package-related problems
When solids form, they often do not just fall straight to the bottom. They may also:
- stick to bottle walls
- collect near caps
- form rings
- deposit in dispensing equipment
- clog filters or nozzles
Easy way to understand it
The precipitated material starts interacting with the package instead of staying in the product.
Why that matters for shelf life
This can cause:
- uneven dosing
- poor redispersion
- unattractive appearance
- residue buildup
- different performance after opening and reuse
For a trainee, this means shelf-life evaluation should include not just the liquid itself, but also what is happening in the package.
10) Precipitation may create a chain reaction of instability
One important lesson in flavor chemistry is that problems often do not stay isolated.
A small amount of precipitation can trigger more instability later.
Easy way to understand it
Once the system starts to fall apart, other problems may follow more easily.
Possible chain reactions
- small crystals seed larger crystals
- emulsion failure becomes oil separation
- oil separation increases oxidation
- oxidation creates off-notes
- off-notes reduce flavor quality
- sediment changes mouthfeel
- mouthfeel changes flavor perception
So precipitation can be the start of a larger aging process, not just one single event.
11) How precipitation affects different kinds of flavor systems
A. Clear beverages
These are often the most sensitive because even small haze or particles are noticeable.
Effects on shelf life:
- visual failure happens early
- top-note loss becomes obvious
- consumers reject the product quickly
B. Cloudy beverages and emulsions
These may hide small instability at first, but precipitation can still change flavor release and aging.
Effects on shelf life:
- separation may worsen over time
- citrus and botanical notes may fade or shift
- oil oxidation risk may increase
C. Syrups and concentrates
These often face crystal growth, especially with sugars, acids, or salts.
Effects on shelf life:
- difficult pouring
- inconsistent dilution
- flavor imbalance in finished use
D. Dairy and plant-protein beverages
These are very sensitive to pH, minerals, and polyphenols.
Effects on shelf life:
- sediment
- graininess
- reduced smoothness
- flavor binding changes
- uneven flavor delivery
E. Botanical and natural extract systems
These often contain many chemically different materials, so precipitation may grow during storage.
Effects on shelf life:
- haze
- dark sediment
- aroma dulling
- stronger bitterness or astringency if profile shifts
12) How trainees should think about shelf-life testing when precipitation is possible
A good trainee should learn that shelf-life testing is not only about whether a product still smells “okay.”
You also need to watch:
- clarity
- haze formation
- sediment formation
- crystal growth
- redispersibility
- ring formation
- flavor uniformity
- aroma loss
- sensory balance over time
Easy way to understand it
Ask two questions during storage:
Does it still look the same?
Does it still taste and smell the same?
If the answer to either question is no, precipitation may be part of the reason.
13) A simple trainee model: how precipitation shortens shelf life
Here is an easy mental model:
Stage 1: Fresh product
Everything is dissolved or well dispersed.
Flavor is balanced.
Appearance is clean.
Aroma is as intended.
Stage 2: Early instability
Tiny particles or small crystals begin forming.
Product may still look acceptable.
Flavor may already begin to drift slightly.
Stage 3: Visible precipitation
Haze, crystals, sediment, or ring appears.
Flavor distribution becomes uneven.
Top notes may weaken or shift.
Stage 4: Advanced aging
The system is no longer uniform.
More oxidation, interaction, or phase separation may occur.
Consumer notices dullness, off-notes, harshness, or inconsistency.
Stage 5: End of acceptable shelf life
The product may still be present physically, but it no longer looks or performs like the intended product.
This is why precipitation is closely tied to practical shelf life.
14) What flavorist trainees should remember most
The most important lesson is this:
Precipitation changes both what the product is and how the product ages.
Once materials fall out of solution, the flavor system is no longer uniform. That can lead to:
- lower flavor strength
- changed sensory balance
- uneven flavor release
- faster oxidative or chemical aging
- poor visual quality
- reduced consumer acceptance
- shorter usable shelf life
So precipitation is not a small side issue. It is one of the important physical changes that can drive product aging.
15) Summary
Precipitation affects flavor aging and shelf life by:
- removing some ingredients from active solution
- changing flavor balance
- reducing aroma intensity
- making flavor release uneven
- promoting further instability
- increasing oxidation risk in some systems
- causing different parts of the product to age differently
- shifting the sensory profile over time
- damaging visual quality
- reducing consumer acceptance
- sometimes interfering with preservatives or functional ingredients
For a trainee, the simplest way to remember it is:
If part of the system falls out, the whole product ages differently.
That is why flavorists monitor precipitation very carefully during stability testing.
###