The Chemistry of Schiff Base Formation: Impact on Flavor, Aging, and Shelf Life
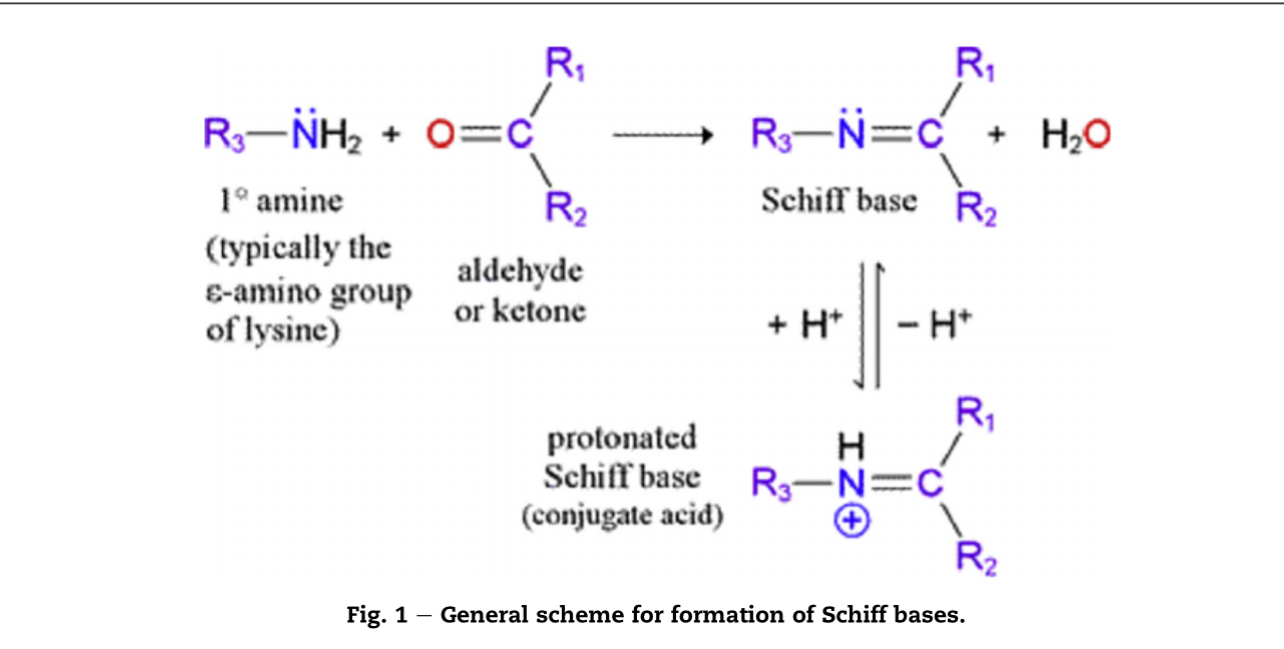
Schiff base formation is a fundamental chemical reaction with profound implications in food science, particularly in the world of flavors. Understanding this process is crucial for formulators aiming to create stable, long-lasting products. This article provides a comprehensive overview of the reaction, the factors that influence it, and its critical role in flavor aging and shelf life.
1) Chemical Groups Involved and Conditions Required
Chemical Groups
Schiff base formation is a specific type of condensation reaction. It occurs between two key functional groups:
- Primary Amine (-NH₂): This group is the nucleophile in the reaction. In food systems, primary amines are commonly found in:
- Amino acids (e.g., lysine, which has a free ε-amino group)
- Proteins and peptides
- Biogenic amines (e.g., putrescine, cadaverine)
- Carbonyl Group (C=O): This group is the electrophile. The most relevant carbonyl compounds in flavor systems are:
- Aldehydes (e.g., vanillin, benzaldehyde, citral)
- Ketones (though they are generally less reactive than aldehydes)
The product of this reaction is an imine, characterized by a carbon-nitrogen double bond (C=N) , which is the Schiff base.
Conditions Required
The reaction is reversible and typically proceeds under relatively mild conditions:
- Solvent: Often occurs in aqueous or hydro-alcoholic systems, which are common in many flavor matrices like beverages, sauces, and creams.
- pH: The reaction is catalyzed by both acids and bases, but the optimal pH range is typically slightly acidic to neutral (pH 4-7) . At very low pH, the amine is protonated (-NH₃⁺) and loses its nucleophilicity. At very high pH, the carbonyl group can undergo side reactions like aldol condensation.
- Temperature: While it can occur at room temperature, the reaction rate increases with temperature, making it a significant factor during processing (e.g., pasteurization, sterilization) and accelerated aging.
2) Factors Accelerating or Inhibiting the Reactions, and Considerations During Formulation
Successfully managing Schiff base formation is a key challenge in flavor formulation. Here are the primary factors to consider.
Factors Accelerating the Reaction
| Factor | Mechanism |
|---|---|
| Higher Temperature | Increases kinetic energy, leading to more frequent and successful collisions between reactants. |
| Optimal pH (4-7) | Provides the ideal balance where the amine is sufficiently unprotonated to react, and the carbonyl carbon is electrophilic enough to be attacked. |
| Water Removal | As a condensation reaction, Schiff base formation releases water. Removing water (e.g., in dry mixes or through evaporation) shifts the equilibrium toward product formation. |
| High Concentration | Increasing the concentration of either the aldehyde or the primary amine increases the reaction rate. |
Factors Inhibiting the Reaction
| Factor | Mechanism |
|---|---|
| Very Low pH (< 3) | The primary amine becomes protonated (-NH₃⁺), making it a poor nucleophile and effectively halting the reaction. |
| Very High pH (> 8) | Aldehydes can undergo base-catalyzed side reactions like Cannizzaro reactions or aldol condensations, depleting the carbonyl reactant. |
| Steric Hindrance | Bulky groups around the amine or carbonyl group can physically impede the necessary close approach of the two reactants. |
| Antioxidants (Indirectly) | By inhibiting the oxidation of precursor molecules (e.g., preventing the oxidation of alcohols to aldehydes), antioxidants can limit the availability of carbonyl reactants. |
Considerations During Formulation
- Ingredient Selection: To minimize browning and off-flavor development, formulators may opt for ingredients with lower free aldehyde content or use encapsulated flavors to physically separate reactive components.
- pH Management: Maintaining a final product pH below 4.0 is a common strategy to inhibit Schiff base formation in beverages and sauces.
- Water Activity (aw): In low-moisture systems, the reaction can be paradoxically accelerated due to the lack of water to drive the reverse reaction. Formulators must consider this in products like powdered drink mixes or dry seasonings.
- Packaging: Using oxygen-barrier packaging helps prevent the oxidative formation of aldehydes from unsaturated fatty acids or alcohols, thereby reducing one of the key reactants over time.
3) Examples of Each Reaction or Process
Here are concrete examples of Schiff base formation in flavor systems.
Example 1: The Classic Vanillin + Lysine Reaction
- Reaction: Vanillin (an aldehyde) reacts with the ε-amino group of the amino acid lysine.
- Outcome: This reaction is responsible for the rapid loss of characteristic vanilla flavor in dairy products, protein shakes, and custards. It also leads to the formation of yellow-brown pigments over time, affecting the product's visual appeal.
Example 2: Citral + Amino Acids in Citrus Beverages
- Reaction: Citral, the key aroma compound in lemongrass and citrus oils, reacts with amino acids present in juice or added as supplements.
- Outcome: This is a major cause of flavor deterioration in citrus-flavored beverages. The reaction depletes the fresh, lemon-like citral and creates off-notes, often described as "terpene-like" or "camphoraceous," while also contributing to haze formation.
Example 3: Maillard Reaction Precursor
- Reaction: The initial step in the Maillard reaction is a Schiff base formed between a reducing sugar (like glucose) and an amino acid.
- Outcome: This is not a degradation pathway but a desired process in cooked, baked, or roasted flavors. The resulting Schiff base rearranges to form the Amadori product, which then breaks down into a vast array of desirable aroma compounds (e.g., pyrazines, furans) and brown melanoidins responsible for the characteristic flavors of coffee, bread, and grilled meat.
4) Impact on Flavor Aging and Shelf Life
The formation of Schiff bases is a double-edged sword in flavor science. While it is essential for creating desirable cooked flavors, it is often a primary driver of undesirable aging.
Negative Impacts on Shelf Life
- Loss of Desired Aroma: The most direct impact is the depletion of aldehyde-based aroma compounds. Fresh, "top-note" flavors like citrus (citral), fruity (hexanal), and vanilla (vanillin) are highly susceptible. Their conversion to odorless or less-desirable Schiff bases leads to a flattening or "muting" of the intended flavor profile.
- Formation of Off-Flavors: In some cases, the Schiff base itself or its subsequent reaction products can generate off-notes. For instance, the reaction with citral can lead to p-cymene and other compounds with undesirable, harsh notes.
- Visual Degradation: The accumulation of conjugated Schiff bases often results in yellowing, browning, or the development of haze in clear liquids. This is a major quality defect in products like clear fruit juices, flavored waters, and cosmetic serums.
- Nutrient Loss: The reaction consumes essential amino acids (like lysine), reducing the nutritional value of protein-fortified products.
Positive Impacts (Controlled Use)
- Development of Cooked Flavors: The Maillard reaction, initiated by a Schiff base, is the cornerstone of creating savory, roasted, and baked notes. This is exploited in creating reaction flavors for bouillons, snacks, and processed meats.
- Antioxidant Properties: Some Schiff bases formed from natural compounds have been shown to exhibit antioxidant activity. In certain contexts, their formation can contribute to the oxidative stability of the product.
- Controlled Release: In advanced formulation, Schiff bases are being explored as a reversible mechanism for controlled release. A flavor aldehyde can be "protected" by forming a Schiff base with a carrier, then released upon a change in pH or upon consumption (e.g., in the mouth) when the equilibrium shifts.
###Conclusion
Schiff base formation is a master variable in flavor stability. For formulators, the key to a successful product lies in understanding and controlling this reaction. By strategically managing pH, ingredient selection, water activity, and processing conditions, it is possible to harness the benefits of this chemistry for creating cooked flavors while mitigating its role in the undesirable aging and degradation of high-value, fresh-flavored products.
Keywords: Schiff base, imine formation, flavor aging, shelf life, flavor stability, Maillard reaction, citral degradation, vanillin, primary amine, aldehyde, food chemistry, formulation
###