Volatilization in Flavor Chemistry: A Complete Practical Guide for Flavorists
The Society of Flavor Chemists requires flavorists to fully understand approximately two dozen reactions and processes that can occur in flavor systems. Flavorists must be able to control these reactions or physical processes to enhance flavor or improve its stability and shelf life. Volatilization is one of the physical processes included among these two dozen reactions and processes.
Introduction
Volatilization is the process by which flavor molecules escape from a product into the air, allowing us to smell and perceive flavor. For trainee flavorists, think of volatilization as the “delivery system” of aroma—if a compound does not volatilize, it cannot be smelled, no matter how strong it is in the formula.
A successful flavor is not just about choosing the right compounds—it’s about controlling how and when they evaporate during processing, storage, and consumption.
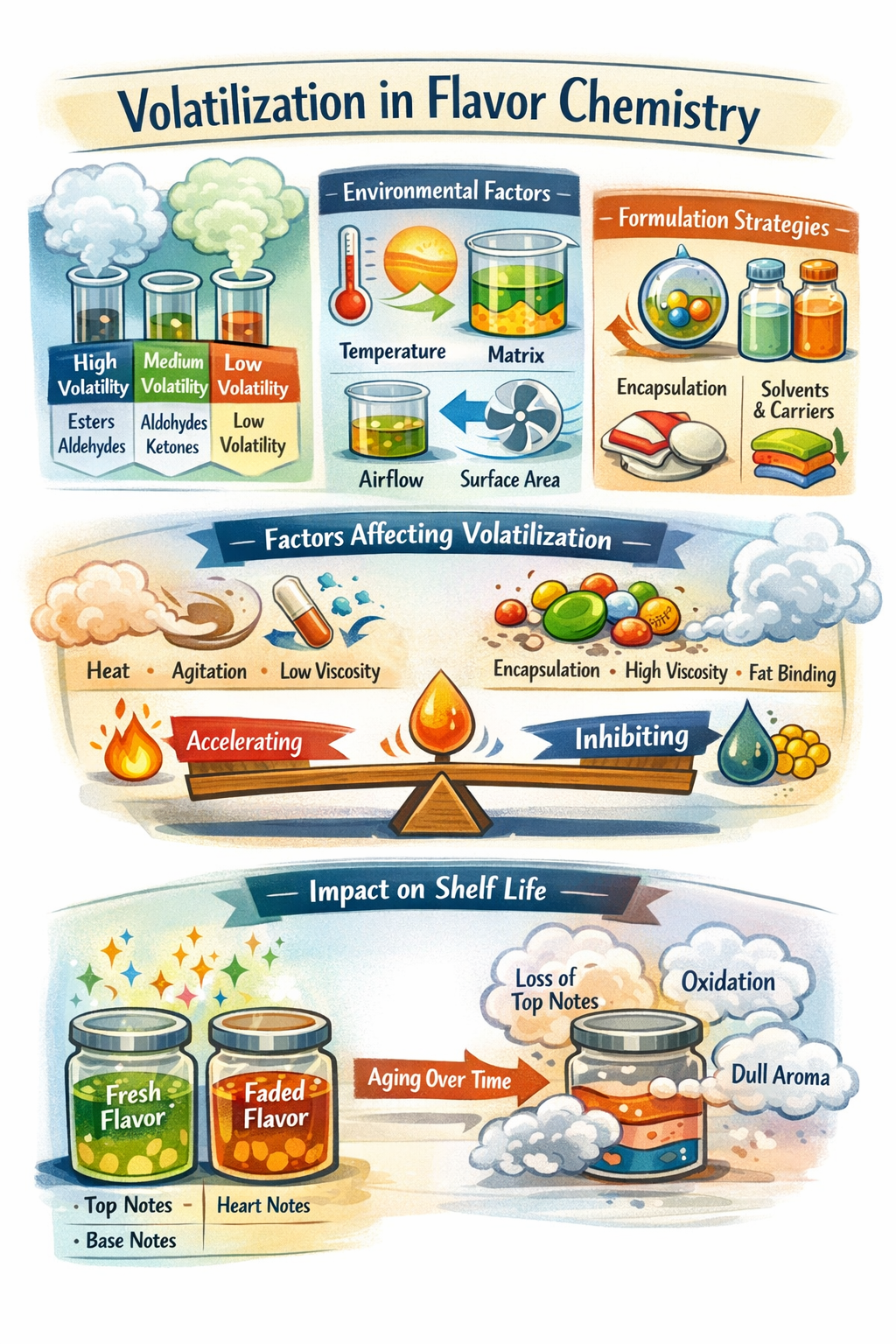
1. Chemical Groups Involved & Conditions Required
A. Key Chemical Groups and Their Volatility Behavior
1. Highly Volatile Compounds (Top Notes)
- Esters (fruity compounds)
These molecules are usually small and light. Because of their size, they escape easily into the air, giving an immediate fruity smell when a product is opened.
→ Example: The strong banana smell when opening candy comes from esters rapidly evaporating. - Aldehydes (green/citrus notes)
Aldehydes often have sharp, fresh odors. They evaporate quickly because they do not strongly stick to other molecules.
→ This is why citrus products smell bright at first but fade quickly. - Low molecular weight ketones
These are also small molecules that require little energy to vaporize, so they contribute to quick aroma impact. - Low molecular weight sulfur compounds
These are extremely potent and volatile. Even tiny amounts can produce strong aromas because they evaporate and reach the nose very easily.
→ Example: Cooked vegetable or savory notes.
Key Traits Explained:
- Low boiling point: They turn into vapor at lower temperatures.
- Weak intermolecular forces: They don’t “stick” to neighboring molecules, so they escape easily.
- Often low polarity: Less interaction with water → easier release into air.
2. Medium Volatility Compounds (Heart Notes)
- Alcohols (e.g., floral compounds)
These form hydrogen bonds with water, so they don’t evaporate as quickly as esters.
→ Result: They last longer and form the “body” of the flavor. - Medium-chain esters
Slightly heavier than top-note esters, so they release more slowly, giving sustained fruity character. - Some ketones and lactones
These contribute creamy or buttery notes and linger longer in the product.
Key Traits Explained:
- Moderate polarity: Some interaction with water slows evaporation.
- Balanced vapor pressure: They neither evaporate too fast nor too slow.
3. Low Volatility Compounds (Base Notes)
- Phenols (smoky, spicy)
These strongly interact with other molecules, so they stay in the product longer. - Lactones (creamy, coconut-like)
Larger molecules that evaporate slowly, contributing to long-lasting flavor. - Long-chain acids and esters
Heavier and less volatile, so they remain even after top notes disappear. - Heavy terpenes
These provide depth and persistence.
Key Traits Explained:
- High molecular weight: Heavier molecules move slower and evaporate less.
- Strong intermolecular interactions: They “stick” to the matrix.
- High boiling point: Require more heat to vaporize.
B. Conditions Required for Volatilization
1. Temperature
- When temperature increases, molecules move faster and escape into the air more easily.
- → Example: Hot soup smells stronger than cold soup because more compounds volatilize.
2. Vapor Pressure
- This is a measure of how easily a compound becomes vapor.
- High vapor pressure = compound prefers to be in the air rather than in the liquid.
- → Trainees can think of this as “willingness to evaporate.”
3. Matrix Composition
- The surrounding ingredients affect how tightly flavor compounds are held.
- → Fat traps hydrophobic compounds, reducing their release.
- → Water allows more release of hydrophilic compounds.
4. Surface Area
- More exposed surface = more molecules can escape at once.
- → Crushed herbs smell stronger than whole herbs.
5. Airflow / Headspace
- Moving air removes volatile molecules from the surface, allowing more to escape.
- → Stirring or shaking increases aroma release.
2. Factors Accelerating or Inhibiting Volatilization & Formulation Considerations
A. Factors Accelerating Volatilization
- High temperature
Heat gives molecules energy to escape faster.
→ Major issue in baking and pasteurization. - Low viscosity systems (thin liquids)
Molecules move freely, so they reach the surface faster.
→ Beverages lose aroma faster than syrups. - Low molecular weight compounds
Smaller molecules require less energy to evaporate. - Low water activity (in some systems)
Less water means fewer interactions holding compounds back. - Air exposure / agitation
Mixing or shaking constantly refreshes the surface, increasing loss. - Ethanol presence
Ethanol can carry volatile compounds into the vapor phase, increasing aroma release.
B. Factors Inhibiting Volatilization
- High viscosity (thick systems)
Thick matrices slow down molecular movement.
→ Example: Syrups retain flavor better than water. - Encapsulation
Flavor compounds are physically trapped and released slowly.
→ Like a “protective shell.” - Fat/oil binding
Lipophilic compounds dissolve in fat and are less likely to escape. - Protein binding
Proteins can hold flavor molecules, reducing release. - Cyclodextrins
These form inclusion complexes, literally trapping molecules inside a ring structure. - Lower temperature
Less energy → slower evaporation.
C. Formulation Considerations for Flavorists
1. Balance Between Release and Retention
- If too volatile → flavor disappears before consumption.
- If too stable → weak aroma perception.
- Goal: Controlled release at the moment of consumption.
2. Use of Carriers and Solvents
- Propylene glycol (PG)
Slows evaporation by holding compounds in solution. - Ethanol
Enhances release, especially in beverages. - Triacetin
Stabilizes heavier notes and reduces rapid loss.
3. Encapsulation Strategies
- Protects sensitive compounds from heat and air.
- Releases flavor gradually during consumption.
4. Matrix Matching
- Beverage: need quick release → use more volatile systems.
- Bakery: need protection → use encapsulated flavors.
- Dairy: consider fat binding effects.
5. Top Note Protection
- Add volatile compounds late in processing.
- Use fixatives to slow their loss.
- Prevent excessive heat exposure.
3. Examples of Volatilization in Real Applications
Example 1: Citrus Beverage
- Citrus oils evaporate quickly, causing flavor fade.
- Emulsions help trap and slowly release these compounds.
Example 2: Baked Goods (Vanilla Flavor)
- Heat drives off volatile compounds during baking.
- Encapsulation protects them until consumption.
Example 3: Chewing Gum
- Requires staged release:
- Immediate burst (top notes)
- Long-lasting flavor (base notes)
Example 4: Savory Snacks
- Sulfur compounds evaporate quickly after processing.
- Reaction flavors help regenerate aroma during eating.
4. Impact on Aging of Flavor and Shelf Life
A. Mechanisms of Flavor Loss
1. Evaporation
- Continuous loss of volatile compounds over time.
- Most noticeable for top notes.
2. Partitioning Changes
- Compounds move into packaging or bind to ingredients.
- Reduces aroma availability.
3. Oxidation + Volatilization
- Oxidation creates new (often undesirable) volatile compounds.
- At the same time, desirable ones are lost.
B. Aging Profile of a Flavor
- Top notes: disappear quickly
- Heart notes: decline gradually
- Base notes: remain longest
→ Result: Flavor becomes dull and less complex.
C. Shelf Life Considerations
1. Packaging
- Good barriers prevent loss of aroma and entry of oxygen.
2. Storage Conditions
- High temperature accelerates volatilization and degradation.
3. Formulation Stability
- Use antioxidants and stabilizers to protect flavor compounds.
D. Practical Shelf-Life Strategy
- Add extra top notes initially to compensate for loss.
- Use layered flavor systems for sustained release.
- Test under accelerated conditions to predict shelf life.
Summary
Volatilization is the key mechanism that controls aroma perception and flavor stability.
Core Concepts for Trainees:
- Small, light molecules evaporate faster and give immediate aroma.
- Temperature, matrix, and formulation determine how fast flavors are released.
- You must balance flavor impact vs. retention.
- Different applications require different volatilization strategies.
- Shelf life is largely determined by how well you control volatilization.
Final Practical Insight
A skilled flavorist does not just create a flavor—they design its behavior over time. Mastering volatilization allows you to build flavors that survive processing, remain stable during storage, and deliver the desired sensory experience exactly when the consumer needs it.
Log in to read more