Crystallization in Flavor Systems: Mechanisms, Matrix Interactions, Risk Control, and Shelf-Life Management
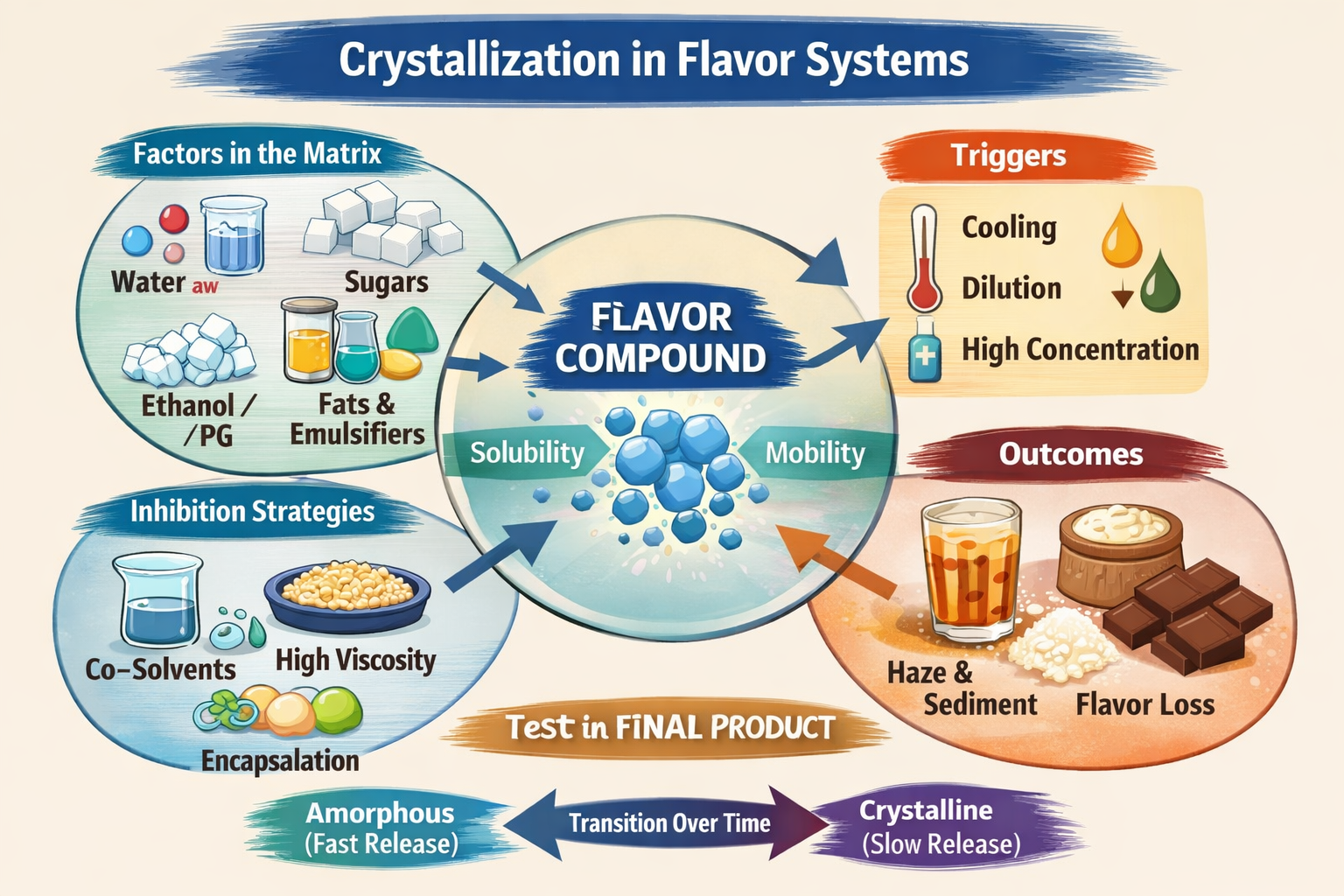
The Society of Flavor Chemists requires flavorists to fully understand approximately two dozen reactions and processes that can occur in flavor systems. Flavorists must be able to control these reactions or physical processes to enhance flavor or improve its stability and shelf life. Crystallization is one of the physical processes included among these two dozen reactions and processes.
Crystallization in Flavor Systems — Explained for Beginners
1. Chemical Groups Involved & Conditions Required
Key Chemical Groups Prone to Crystallization
• Hydroxyl groups (–OH)
👉 What this means:
These are groups found in sugars and polyols (like sorbitol).
👉 Why they crystallize easily:
Hydroxyl groups can form hydrogen bonds, which act like tiny “magnets” between molecules. These interactions help molecules line up neatly into crystals.
👉 Simple analogy:
Think of Lego bricks with strong connectors—they easily snap into a structured shape.
👉 Flavorist takeaway:
Sugars and polyols often crystallize → expect issues like grittiness or caking.
• Carboxylic acids (–COOH)
👉 What this means:
Found in acids like citric acid, malic acid, tartaric acid.
👉 Why they crystallize:
They are highly polar and strongly attract each other, forming very stable crystal structures.
👉 Simple analogy:
Like magnets snapping tightly into a fixed grid.
👉 Flavorist takeaway:
Acidulants can form visible crystals or sediment if the system becomes unstable.
• Aldehydes & phenols
👉 What this means:
Includes compounds like vanillin.
👉 Why they crystallize:
They are structured and somewhat polar, allowing them to stack together neatly.
👉 Simple analogy:
Flat cards stacking into a tidy deck.
👉 Flavorist takeaway:
Vanillin crystallization is one of the most common real-world problems in flavors.
• Esters (some)
👉 What this means:
Most esters are liquids, but some heavier or symmetrical ones can crystallize.
👉 Why they sometimes crystallize:
If they are symmetrical and pure, they can pack together like solids.
👉 Flavorist takeaway:
Usually low risk, but watch heavy or high-purity esters in cold systems.
• Lipids (triglycerides)
👉 What this means:
Fats and oils used as carriers or in food systems.
👉 Why they crystallize:
They form different crystal structures (polymorphs) depending on temperature.
👉 Simple analogy:
Like water forming ice in different shapes depending on how it freezes.
👉 Flavorist takeaway:
Fat crystallization affects texture, mouthfeel, and flavor release.
Conditions Required for Crystallization
• Supersaturation
👉 What this means:
More material is dissolved than the liquid can normally hold.
👉 Why it matters:
This is the starting point for crystallization—without it, crystals won’t form.
👉 Simple analogy:
Dissolving too much sugar in hot water—when it cools, crystals form.
👉 Flavorist takeaway:
High loading of flavor compounds increases crystallization risk.
• Nucleation & crystal growth
Nucleation
👉 What this means:
The first tiny crystal forms.
👉 Why it matters:
Once one crystal forms, others follow quickly.
👉 Analogy:
Like the first snowflake starting a snowfall.
Crystal growth
👉 What this means:
More molecules attach to the initial crystal.
👉 Why it matters:
Small crystals become visible particles or sediment.
👉 Flavorist takeaway:
Preventing nucleation is often easier than stopping growth later.
• Temperature drop
👉 What this means:
Cooling reduces solubility.
👉 Why it matters:
Compounds that were dissolved at warm temperatures may fall out as crystals when cooled.
👉 Flavorist takeaway:
Always test cold stability (fridge conditions).
• Solvent polarity changes
👉 What this means:
Changing from alcohol-rich to water-rich systems.
👉 Why it matters:
Some compounds dissolve well in alcohol but not in water.
👉 Example:
A flavor concentrate is clear → becomes cloudy in a beverage.
👉 Flavorist takeaway:
Dilution into final product is a major crystallization trigger.
• Concentration increase
👉 What this means:
More compound per volume.
👉 Why it matters:
Higher chance of reaching supersaturation.
👉 Flavorist takeaway:
Overloading actives (e.g., vanillin, menthol) causes instability.
• Agitation (mixing)
👉 Low agitation: fewer crystals, but larger
👉 High agitation: more crystals, but smaller
👉 Why it matters:
Mixing affects crystal size and appearance.
👉 Flavorist takeaway:
In beverages, many small crystals = haze; large crystals = sediment.
2. Factors Accelerating or Inhibiting Crystallization
Factors That Accelerate Crystallization
• High supersaturation
👉 More “extra” material → stronger push to form crystals.
👉 Takeaway:
High loading = high risk.
• Presence of seed crystals
👉 Even tiny dust particles or undissolved solids can start crystallization.
👉 Analogy:
Like a tiny ice crystal turning water into ice quickly.
👉 Takeaway:
Filtration and clean processing matter.
• Low temperature
👉 Reduces solubility → compounds come out of solution.
👉 Takeaway:
Cold chain = critical test condition.
• Low viscosity systems
👉 Molecules move freely → easier to organize into crystals.
👉 Takeaway:
Thin beverages are more at risk than thick syrups.
• High purity
👉 Pure compounds align more easily into crystals.
👉 Takeaway:
Ironically, very pure materials can be less stable physically.
Factors That Inhibit Crystallization
• High viscosity
👉 Thick systems slow molecular movement.
👉 Analogy:
Like trying to build a structure in honey vs water.
👉 Takeaway:
Gums and syrups can help prevent crystallization.
• Impurities / complex mixtures
👉 Mixed molecules disrupt crystal formation.
👉 Takeaway:
Blends are often more stable than single compounds.
• Emulsifiers & stabilizers
👉 They block crystal growth surfaces.
👉 Takeaway:
Useful tools in beverages and emulsions.
• Rapid cooling
👉 Molecules don’t have time to form crystals → become amorphous.
👉 Takeaway:
Spray drying uses this principle.
• Encapsulation
👉 Traps compounds in a matrix.
👉 Takeaway:
Improves stability and shelf life.
Formulation Considerations
• Beverages
👉 Problem: haze, sediment
👉 Solution: co-solvents, emulsifiers
👉 Takeaway:
Clarity = key quality parameter.
• Powder flavors
👉 Problem: caking
👉 Sometimes crystals help flow
👉 Takeaway:
Balance between flowability and stability.
• Confectionery
👉 Controlled crystallization = good texture
👉 Uncontrolled = gritty
👉 Takeaway:
Crystallization can be designed intentionally.
• Encapsulated flavors
👉 Prefer amorphous state
👉 Takeaway:
Better flavor release and stability.
3. Examples of Crystallization
• Vanillin crystallization
👉 Crystals appear in concentrates or finished products
👉 Effect: weaker aroma release
👉 Takeaway:
One of the most common troubleshooting cases.
• Citric acid crystallization
👉 Appears as sediment
👉 Takeaway:
Often triggered by temperature or water activity changes.
• Menthol crystallization
👉 Naturally forms crystals
👉 Takeaway:
Must be carefully dissolved and tested cold.
• Sugar crystallization
👉 Can be good (smooth fondant) or bad (gritty texture)
👉 Takeaway:
Control is everything.
• Fat crystallization
👉 Affects mouthfeel and release
👉 Takeaway:
Important in dairy, chocolate, and encapsulated systems.
4. Impact on Aging & Shelf Life
• Reduced flavor release
👉 Crystals dissolve slower
👉 Takeaway:
Top notes become weaker over time.
• Physical instability
👉 Cloudiness, sediment
👉 Takeaway:
Consumers see this as a defect.
• Loss of homogeneity
👉 Flavor unevenly distributed
👉 Takeaway:
Inconsistent taste experience.
• Powder caking
👉 Crystals bind particles together
👉 Takeaway:
Poor handling and dosing.
• Fat polymorphism
👉 Texture changes over time
👉 Takeaway:
Can impact both texture and flavor perception.
• Shelf life reduction mechanisms
Amorphous → crystalline transition
👉 Releases trapped flavor
👉 Takeaway:
Leads to flavor loss over time.
Water migration
👉 Triggers crystallization
👉 Takeaway:
Moisture control is critical.
Temperature cycling
👉 Repeated dissolve/recrystallize
👉 Takeaway:
Shipping conditions matter.
Final Summary (Beginner Perspective)
Crystallization happens when molecules find a stable, organized structure—and many flavor compounds naturally want to do this.
For a flavorist, the key ideas are:
- Some ingredients (vanillin, menthol, acids, sugars) are naturally crystal-formers
- Crystallization is triggered by:
- cooling
- water changes
- high concentration
- It leads to:
- haze
- sediment
- weaker flavor
- texture defects
- But it can also be useful when controlled (e.g., confectionery)
👉 The real skill is:
not just avoiding crystallization—but controlling it depending on the product.
How Ingredients in a Food Matrix Affect Crystallization of a Specific Flavor Compound
Introduction
Crystallization rarely depends on just one compound alone. In real products, a flavor compound sits inside a complex food matrix—water, sugars, acids, solvents, fats, proteins, and stabilizers all interacting at once.
👉 Think of it this way:
A compound may be perfectly stable in a flavor bottle, but once added to a beverage, dairy product, or powder system, it can suddenly crystallize, haze, or sediment.
So the key question is not just “Does this compound crystallize?”
👉 It is: “How does the surrounding matrix push it toward or away from crystallization?”
1. Role of Water (the Most Important Factor)
• Water reduces solubility for many flavor compounds
👉 Many flavor compounds (like vanillin or menthol) dissolve better in alcohol or oil than in water.
👉 When a flavor is diluted into a beverage:
- ethanol ↓
- water ↑
→ compound becomes less soluble → crystallization risk ↑
👉 Example:
A clear vanilla flavor concentrate becomes cloudy after dilution into a soft drink.
• Water activity (aw) controls crystallization behavior
👉 Not just how much water, but how “free” the water is.
- Low aw → favors crystallization of sugars/acids
- High aw → promotes dissolution
👉 Example:
Powdered drink mixes may crystallize during storage if moisture slowly migrates.
• Water migration triggers crystallization
👉 Water moving within a product can locally change concentration.
👉 Example:
In filled confections:
- water moves from moist filling → dry shell
→ sugar crystallizes at interface → gritty texture
2. Role of Solvents (Ethanol, Propylene Glycol, Glycerin)
• Co-solvents increase solubility
👉 Ethanol and propylene glycol help dissolve many flavor compounds.
👉 Why:
They reduce polarity mismatch between compound and water.
👉 Example:
Menthol stays dissolved in ethanol-rich flavor but crystallizes when diluted into water.
• Solvent loss triggers crystallization
👉 When solvent evaporates or is diluted:
- solubility ↓
- supersaturation ↑
→ crystals form
👉 Example:
Flavor applied to a snack → solvent evaporates → crystals form on surface.
• Solvent balance is critical
👉 Too little solvent → crystallization
👉 Too much solvent → regulatory or sensory issues
👉 Flavorist takeaway:
Always optimize solvent ratio, not just ingredient levels.
3. Role of Sugars and Polyols
• Sugars can compete for water
👉 Sugars like sucrose bind water strongly.
👉 This leaves less “free water” for dissolving other compounds.
👉 Result:
Flavor compound crystallization risk increases.
• Sugars increase viscosity
👉 Thick systems slow molecule movement.
👉 Effect:
- slows crystal formation
- but once formed, crystals may persist
• Sugars themselves crystallize
👉 They can act as seed crystals.
👉 Example:
Sugar crystallization can trigger other compounds to crystallize (co-crystallization effect).
• Polyols (e.g., sorbitol, erythritol)
👉 Can either:
- stabilize (by holding water)
- or crystallize themselves
👉 Example:
Erythritol crystallization causes cooling and gritty texture in reduced-sugar systems.
4. Role of Acids and pH
• pH affects solubility
👉 Some compounds change form depending on pH.
👉 Example:
Acids like citric acid:
- more soluble at certain pH
- may crystallize if pH shifts
• Acid–salt balance
👉 Converting acid → salt form often increases solubility.
👉 Example:
Sodium citrate dissolves better than citric acid.
• High acid systems
👉 Can:
- increase ionic strength
- reduce solubility of some organics
👉 Result:
Unexpected crystallization in acidic beverages.
5. Role of Fats and Oils
• Lipophilic compounds dissolve better in fats
👉 Compounds prefer environments similar to themselves.
👉 Example:
Menthol is more stable in oil than in water.
• Fat crystallization traps or releases flavors
👉 When fats crystallize:
- they can trap flavor compounds
- or push them out (phase separation)
• Polymorphism matters
👉 Different fat crystal forms release flavor differently.
👉 Example:
Chocolate bloom affects flavor release and texture.
6. Role of Emulsifiers and Surfactants
• Improve solubility via micelles
👉 Emulsifiers surround hydrophobic molecules.
👉 Effect:
Prevents them from coming together and forming crystals.
• Inhibit crystal growth
👉 They block crystal surfaces.
👉 Analogy:
Like putting barriers around growing crystals.
• Stabilize emulsions
👉 Keeps flavor distributed evenly.
👉 Example:
Cloudy beverages rely on emulsifiers to prevent oil-phase separation and crystallization.
7. Role of Proteins and Hydrocolloids
• Increase viscosity
👉 Slows down molecular movement.
👉 Effect:
Reduces crystallization rate.
• Interact with flavor molecules
👉 Proteins may bind flavor compounds.
👉 Result:
Less free compound → less crystallization.
• Hydrocolloids (gums)
👉 Examples: xanthan gum, gum arabic
👉 Effects:
- stabilize system
- reduce nucleation
- control water mobility
👉 Flavorist takeaway:
Very powerful tools for stabilization.
8. Role of Temperature and Storage Conditions
• Cooling reduces solubility
👉 Most compounds dissolve less at low temperature.
👉 Example:
Clear beverage becomes cloudy in refrigerator.
• Temperature cycling
👉 Repeated heating/cooling:
- dissolve → recrystallize → grow crystals
👉 Result:
Larger, visible crystals over time.
• Freezing
👉 Ice formation concentrates solutes.
👉 Result:
Crystallization of flavor compounds increases.
9. Role of Impurities and Matrix Complexity
• Impurities disrupt crystallization
👉 Mixed systems make it harder for molecules to align.
👉 Effect:
More stable (less crystallization).
• But can also cause instability
👉 Some impurities act as nucleation sites.
👉 Result:
Unexpected crystallization.
10. Real-World Example (Putting It All Together)
Case: Vanillin in a Beverage
- In flavor concentrate:
- dissolved in ethanol → stable
- After dilution:
- water ↑
- ethanol ↓
- temperature ↓
👉 Result:
- solubility drops
- supersaturation occurs
- vanillin crystallizes → haze/sediment
👉 Fix:
- increase co-solvent
- reduce vanillin loading
- add stabilizer
Key Takeaways for Flavorists
👉 Crystallization is not just about the compound—it’s about the environment around it.
Most important drivers:
- Water content and activity
- Solvent system
- Temperature
- Concentration
- Presence of other ingredients
Golden Rules
- Always test in final matrix, not just concentrate
- Cold stability testing is essential
- Watch solvent dilution effects
- Control water activity in powders
- Use stabilizers strategically
Summary
Ingredients in a food matrix affect crystallization by changing:
- how soluble a compound is
- how fast molecules can move
- whether crystals can start forming
A compound that is stable in one system may crystallize in another because:
- water increases
- solvent decreases
- temperature drops
- other ingredients interfere
👉 The real expertise in flavor formulation is understanding these interactions and designing the matrix to keep compounds stable—or to crystallize only when desired.
Advanced Recommendations on Crystallization for Flavorists (What Professionals Know That Beginners Often Miss)
1. Treat Crystallization as a Design Parameter, Not Just a Problem
Most beginners try to eliminate crystallization completely. That’s not always the right approach.
👉 In reality, crystallization can be:
- Beneficial → texture in confectionery, controlled release systems
- Neutral → stable powders
- Harmful → haze, sediment, caking
👉 Professional mindset:
Instead of asking “How do I stop crystallization?”
Ask → “Where, when, and how much crystallization do I want?”
2. Always Think in Terms of “Solubility Margin” (Not Just Solubility)
What beginners miss:
They check if a compound dissolves… but not how close it is to failing.
👉 Solubility margin = how far below the limit you are
- Large margin → stable
- Small margin → high risk
👉 Example:
A flavor that is 95% of solubility limit may look perfect…
…but will crystallize with:
- slight cooling
- minor evaporation
- small formulation change
👉 Recommendation:
Always build at least a 20–50% safety margin for critical compounds like:
- vanillin
- menthol
- citric acid
3. Learn to Recognize Early Warning Signs
Before visible crystals appear, systems often show:
- slight haze
- “oily” streaks
- viscosity changes
- delayed precipitation (hours or days later)
👉 Key insight:
Crystallization is often slow and silent at first.
👉 Recommendation:
- Check samples after 24–72 hours, not just immediately
- Use simple tools:
- flashlight (for haze)
- microscope (optional but powerful)
4. Understand the Difference Between These Three (Very Important)
• Crystallization
Ordered solid structure
→ sharp particles, sediment
• Precipitation
General term (may or may not be crystalline)
• Phase separation
Oil/water separation (not crystals)
👉 Why this matters:
Each has different solutions:
- crystallization → adjust solubility
- precipitation → adjust chemistry
- phase separation → adjust emulsification
👉 Common mistake:
Trying to fix crystallization with emulsifiers (won’t work).
5. Master “Temperature Abuse” Testing
Real products don’t stay at one temperature.
👉 They experience:
- refrigeration
- room temperature
- hot transport
- freeze–thaw cycles
👉 Recommendation: Always test:
- 4°C (refrigerator)
- 25°C (room temp)
- 40°C (accelerated)
- freeze–thaw (at least 2–3 cycles)
👉 Why:
Crystallization often appears only after temperature cycling, not at constant temperature.
6. Use “Anti-Crystallization Strategies Toolbox”
Instead of guessing, think in categories:
A. Solubility strategies
- add ethanol or propylene glycol
- reduce loading
- change compound form (salt vs acid)
B. Mobility control
- increase viscosity (gums, syrups)
- reduce molecular movement
C. Nucleation control
- filtration
- avoid dust/particles
- smooth processing
D. Crystal growth inhibition
- emulsifiers
- mixed systems (impurities help)
👉 Key insight:
You don’t need to eliminate crystallization completely—just slow it enough for shelf life.
7. Beware of “Hidden Crystallizers” in Your System
Not all problems come from flavor compounds
Watch for:
- sugars (e.g., sucrose)
- polyols (erythritol, sorbitol)
- acids
- salts
👉 These can:
- crystallize themselves
- trigger crystallization of other compounds
👉 Example:
Sugar crystals can act as seeds for vanillin crystallization.
8. Understand Amorphous vs Crystalline States
Crystalline
- stable
- slow dissolution
- lower flavor release
Amorphous
- unstable
- fast dissolution
- better flavor release
👉 Key concept:
Many encapsulated flavors are intentionally amorphous.
👉 Risk:
They may slowly crystallize over time → flavor loss.
9. Pay Attention to Particle Size (Often Overlooked)
👉 Smaller crystals:
- look like haze
- dissolve faster
👉 Larger crystals:
- visible sediment
- gritty texture
👉 Recommendation:
- control nucleation to influence particle size
- sometimes many tiny crystals are better than a few large ones
10. Regulatory & Labeling Considerations
Crystallization can indirectly affect compliance:
- sediment may be seen as quality defect
- instability may shorten shelf life claims
- re-dissolution issues affect consumer usage
👉 Especially critical in:
- clear beverages
- premium products
- clean-label systems (fewer stabilizers)
11. Build a Personal “Crystallization Memory”
Experienced flavorists rely heavily on memory:
👉 You should track:
- which compounds crystallize easily
- which solvent systems work
- which applications fail
👉 Recommendation:
Create your own:
- “high-risk compound list”
- “safe solvent ratios”
- “failure case log”
12. The Most Important Practical Rule
👉 Always test in the final product—not just the flavor concentrate
Because:
- water changes everything
- pH changes everything
- other ingredients interfere
👉 A flavor that is perfect in the lab can fail instantly in:
- beverage
- dairy
- powder
- frozen product
Final Summary
To truly master crystallization, a flavorist must go beyond theory and think like this:
- It is not just chemistry → it is system behavior
- It is not instant → it is time-dependent
- It is not isolated → it is matrix-driven
Professional-level mindset:
- control solubility margin
- anticipate temperature effects
- manage water and solvents
- use stabilization strategies intentionally
- test under real-world conditions
👉 Ultimately, crystallization control is one of the key differences between:
- a lab formula that looks good
- and a commercial product that survives shelf life
50+ Compound Crystallization Risk Library for Flavorists
This library is meant as a practical screening tool for flavor development. It helps answer a simple question:
Which compounds are more likely to crystallize out of a flavor or finished food system, and under what circumstances?
Crystallization risk depends on several things working together:
- the chemical structure of the molecule
- its melting point
- its solubility in the carrier system
- the purity of the material
- the storage temperature
- the presence of water, ethanol, propylene glycol, oils, gums, sugars, or salts
In general, compounds with higher melting points, stronger intermolecular attraction, lower solubility, and more rigid/symmetrical structures have higher crystallization risk.
To make this useful, each entry includes:
- Compound
- Main flavor use / note
- Crystallization risk: Low / Moderate / High / Very high
- Why
- Formulation caution
How to read the risk levels
Low
Usually remains liquid or well dissolved under normal flavor handling conditions.
Moderate
Can crystallize under stress: cold storage, high loading, solvent shift, water pickup, or long storage.
High
Frequently crystallizes unless carefully managed with solvent balance, dilution, or temperature control.
Very high
Commonly encountered as crystals or solids; strong tendency to crystallize in concentrates or cold conditions.
A. Sweet, Vanilla, Phenolic, and Balsamic Materials
1. Vanillin
Use: vanilla, creamy, sweet
Risk: Very high
Why: relatively rigid aromatic structure, fairly high melting point, strong packing ability
Formulation caution: often crystallizes in concentrates, especially in low-solvent or cold systems; watch in clear beverages and vanilla emulsions.
2. Ethyl vanillin
Use: stronger vanilla note
Risk: Very high
Why: even stronger crystal-forming tendency than vanillin in many systems because of high potency and solid character
Formulation caution: easy to overload because use levels are low but concentrate levels can still exceed solubility.
3. Maltol
Use: sweet, caramelized, cotton-candy, jammy
Risk: High
Why: polar heterocycle with strong intermolecular attraction
Formulation caution: can crystallize in syrups, powdered bases, and concentrated sweet systems.
4. Ethyl maltol
Use: candy, caramelized, cotton-candy
Risk: High
Why: solid, relatively high melting point, limited solubility in some systems
Formulation caution: frequently problematic in concentrates and clear beverages if not pre-dissolved well.
5. p-Hydroxybenzaldehyde
Use: phenolic, sweet, anisic support
Risk: High
Why: aromatic aldehyde with polar functionality and strong crystal packing
Formulation caution: often better handled in solvent blends than in water-heavy systems.
6. Piperonal
Use: heliotrope, vanilla-floral, cherry-almond support
Risk: High
Why: rigid aromatic structure and solid nature
Formulation caution: may crystallize in perfume-like flavor topnotes and confectionery bases if overloaded.
7. Coumarin
Use: tonka-like, sweet hay note; restricted or not permitted in many food uses depending on jurisdiction
Risk: High
Why: planar aromatic lactone, solid crystal-former
Formulation caution: from a physical standpoint it crystallizes readily; from a regulatory standpoint it also requires great caution.
8. Zingerone
Use: ginger, spicy-sweet, warm
Risk: Moderate to high
Why: phenolic ketone with solid character
Formulation caution: can drop out in spicy concentrates and low-polarity systems.
9. Eugenol
Use: clove, spicy, phenolic
Risk: Low to moderate
Why: liquid at normal room conditions, but may associate strongly and become less mobile at low temperature
Formulation caution: usually not a major crystallization problem by itself, but can contribute to clouding in stressed systems.
10. Isoeugenol
Use: clove-spicy-carnation
Risk: Moderate
Why: similar to eugenol but may behave less forgivingly depending on purity and temperature
Formulation caution: monitor in cold storage and in solvent-lean systems.
B. Cooling, Mint, and Terpenic Alcohols
11. Menthol
Use: cooling, mint
Risk: Very high
Why: classic crystal-forming flavor material; solid at room temperature in pure form
Formulation caution: one of the most common crystallization issues in mint flavors, oral care flavors, and beverage compounds.
12. L-Menthol crystals in blends
Use: cooling agent
Risk: Very high
Why: even when dissolved warm, it often recrystallizes on cooling
Formulation caution: always check cold-room and freezer stability.
13. Isomenthol
Use: mint/cooling support
Risk: High
Why: menthol-family solid behavior
Formulation caution: less commonly used, but still crystallization-prone.
14. Borneol
Use: camphoraceous, herbal
Risk: High
Why: bicyclic alcohol with rigid structure and strong packing tendency
Formulation caution: may crystallize in herbal and pine profiles.
15. Camphor
Use: camphoraceous, medicinal
Risk: High
Why: rigid bicyclic ketone with strong tendency to form crystals
Formulation caution: can form visible solids in cold storage.
16. Terpineol
Use: lilac, pine, floral, terpene support
Risk: Moderate
Why: less severe than menthol, but some grades or isomer distributions may haze or partly crystallize
Formulation caution: verify with winterization testing.
17. Thymol
Use: thyme, medicinal, phenolic
Risk: High
Why: phenolic terpene with solid tendency
Formulation caution: watch especially in savory herbal concentrates.
18. Carvacrol
Use: oregano, phenolic herbal
Risk: Moderate
Why: often liquid, but can show low-temperature solidification behavior depending on purity
Formulation caution: less severe than thymol, but still monitor in cold chain products.
C. Organic Acids and Acidulants
19. Citric acid
Use: sourness, acidulation
Risk: High
Why: strong hydrogen bonding, clear crystal-forming acid
Formulation caution: if dissolved near saturation, temperature drop or water loss can cause crystallization.
20. Malic acid
Use: smooth sourness, fruit acidity
Risk: High
Why: dicarboxylic acid with strong lattice formation
Formulation caution: common issue in concentrated beverage systems and powdered applications.
21. Tartaric acid
Use: grape, wine, sharp acidity
Risk: High
Why: highly polar acid, readily crystalline
Formulation caution: especially important in grape, wine, and hard candy systems.
22. Fumaric acid
Use: persistent sourness, dry beverage systems
Risk: Very high
Why: very low water solubility relative to many common acids
Formulation caution: excellent for some dry systems precisely because it stays undissolved longer, but in liquids it is a strong precipitation/crystallization risk.
23. Succinic acid
Use: savory, acid support, shellfish/meaty nuance
Risk: High
Why: dicarboxylic acid with strong crystal tendency
Formulation caution: watch in savory concentrates and low-water systems.
24. Adipic acid
Use: acidulant in dry applications
Risk: High
Why: low-to-moderate solubility and clear crystalline nature
Formulation caution: often acceptable in powders, but problematic in beverages if not well managed.
25. Lactic acid
Use: acidulant, dairy nuance
Risk: Low
Why: usually supplied as aqueous syrup or liquid; much less likely to crystallize than dry organic acids
Formulation caution: water activity changes matter more than classic crystal drop-out.
26. Benzoic acid
Use: preservative-associated acidic aromatic note context
Risk: High
Why: aromatic acid with limited solubility
Formulation caution: solvent choice matters greatly.
27. Sorbic acid
Use: preservative context
Risk: Moderate to high
Why: limited solubility and solid behavior
Formulation caution: in formulation work it may deposit if the medium changes.
D. Sugars, Polyols, and Sweetness Modulators
28. Sucrose
Use: sweetener, bulking
Risk: Very high
Why: classic crystal former
Formulation caution: desired in some products, disastrous in others; uncontrolled crystallization causes grittiness.
29. Glucose
Use: sweetener, syrup solids
Risk: High
Why: crystallizes readily depending on water content and temperature
Formulation caution: key issue in confectionery and syrup stability.
30. Fructose
Use: sweetener
Risk: Moderate
Why: usually more hygroscopic and often less eager to crystallize than sucrose under many conditions
Formulation caution: still can crystallize in concentrated systems.
31. Dextrose monohydrate
Use: sweetener, bulking
Risk: Very high
Why: highly crystalline material
Formulation caution: contributes strongly to caking and gritty texture if moisture shifts.
32. Sorbitol
Use: sweetener, humectant
Risk: High
Why: can crystallize from concentrated solutions or during long storage
Formulation caution: sorbitol crystallization is a major texture issue in soft confections and reduced-sugar products.
33. Mannitol
Use: cooling sweetener, powder systems
Risk: Very high
Why: strong crystal-forming polyol
Formulation caution: excellent where crystalline structure is desired; risky where smooth dissolution is needed.
34. Xylitol
Use: cooling sweetener
Risk: High
Why: readily crystalline polyol
Formulation caution: widely used, but can recrystallize and alter mouthfeel.
35. Erythritol
Use: bulk sweetener
Risk: Very high
Why: notorious for crystallization and cooling effect
Formulation caution: one of the most common reduced-sugar texture problems.
36. Lactose
Use: dairy solids context
Risk: High
Why: limited solubility and crystallization tendency
Formulation caution: especially important in dairy powders, fillings, and milk-based flavors.
E. Amino Acids, Nitrogen Compounds, and Savory Components
37. Glycine
Use: sweetness, buffering, savory reaction systems
Risk: High
Why: simple, highly regular amino acid; good crystal former
Formulation caution: can crystallize in concentrated reaction flavor intermediates.
38. Alanine
Use: sweet-savory support
Risk: High
Why: amino acid crystal behavior
Formulation caution: most relevant in dry blends and concentrated process flavors.
39. Monosodium glutamate (MSG)
Use: umami
Risk: High
Why: ionic solid, clearly crystalline
Formulation caution: usually intentionally used as crystals or granules, but in liquids it can precipitate/crystallize if overloaded.
40. Disodium inosinate
Use: umami enhancer
Risk: Moderate to high
Why: salt-like behavior and solubility dependence
Formulation caution: monitor in concentrated savory bases.
41. Disodium guanylate
Use: umami enhancer
Risk: Moderate to high
Why: similar to inosinate
Formulation caution: solvent and salt balance matter.
42. Creatine / creatinine related savory systems
Use: meat reaction systems
Risk: Moderate
Why: can contribute to solid formation in concentrated reaction mixtures
Formulation caution: often part of complex matrices, so behavior is system-dependent.
F. Lactones, Furans, and Maillard-Type Sweet-Brown Materials
43. Furaneol
Use: strawberry, caramelized, cotton-candy, jam
Risk: Moderate to high
Why: polar, solid character, can crystallize in concentrates
Formulation caution: potency is high, but so is physical instability in some systems.
44. Sotolon
Use: maple, curry, fenugreek, aged note
Risk: Moderate
Why: can be stable at low use, but concentrated intermediates may show crystallization issues
Formulation caution: less common as a visible crystal problem because use level is low.
45. Gamma-undecalactone
Use: peach, apricot
Risk: Low to moderate
Why: usually oily/liquid behavior, but can partly solidify under cold conditions depending on purity
Formulation caution: watch in refrigerated emulsions.
46. Gamma-decalactone
Use: peach, creamy fruit
Risk: Low to moderate
Why: generally manageable, but cold hazing may happen
Formulation caution: not a classic severe crystal former, but worth checking in clear systems.
47. Delta-decalactone
Use: creamy, coconut, dairy
Risk: Moderate
Why: can show partial solidification in cool conditions
Formulation caution: may create cloud or waxy deposition in beverage emulsions.
48. Massoia lactone
Use: creamy coconut
Risk: Moderate
Why: some lactones become less soluble in cooler or more aqueous systems
Formulation caution: evaluate in finished matrix, not just in the concentrate.
G. Esters and Fruity Materials That Can Solidify or Crystallize
Most common low-molecular esters are not major crystal risks because they are liquids. But some higher molecular weight or purer materials can show cold crystallization or solidification.
49. Benzyl acetate
Use: jasmine, fruity, floral
Risk: Low
Why: usually liquid and manageable
Formulation caution: not usually a crystallization concern.
50. Benzyl benzoate
Use: balsamic, solvent-like carrier, floral
Risk: Moderate
Why: relatively heavy aromatic ester; can become sluggish or partly solidify cold
Formulation caution: check in winter conditions.
51. Phenethyl alcohol
Use: rose, floral
Risk: Low to moderate
Why: usually liquid but can become less mobile at low temperature
Formulation caution: more clouding than classic hard crystals in many systems.
52. Phenethyl acetate
Use: honey, floral, fruity
Risk: Low
Why: generally liquid and stable
Formulation caution: low concern.
53. Dibenzyl ether / heavy floral carriers
Use: floral support
Risk: Moderate
Why: heavier aromatic materials may form solids or haze on chilling
Formulation caution: system-dependent.
H. Spicy, Warm, and Aromatic Aldehydes / Ketones
54. Cinnamic acid
Use: cinnamon-related support
Risk: High
Why: aromatic acid, strong crystal former
Formulation caution: much more crystal-prone than cinnamaldehyde.
55. Cinnamaldehyde
Use: cinnamon
Risk: Low to moderate
Why: usually liquid, though it can participate in incompatibility or polymerization issues more than crystallization
Formulation caution: crystallization is usually not the main issue, but cold viscosity changes can confuse diagnosis.
56. Anisic aldehyde
Use: hawthorn, anisic, sweet floral
Risk: Moderate to high
Why: aromatic aldehyde with solid tendency
Formulation caution: can crystallize from concentrates.
57. Acetovanillone
Use: vanilla, creamy phenolic support
Risk: High
Why: aromatic ketone with solid nature
Formulation caution: often treated similarly to other phenolic solids.
I. Salts, Buffers, and Functional Components Often Present with Flavors
58. Sodium citrate
Use: buffer, acid balance
Risk: Moderate to high
Why: ionic solid with finite solubility
Formulation caution: less troublesome than citric acid in some systems, but still can deposit.
59. Potassium sorbate
Use: preservative
Risk: Moderate
Why: salt form improves solubility versus sorbic acid, but concentrated systems can still precipitate solids
Formulation caution: pH and solvent balance matter.
60. Sodium benzoate
Use: preservative
Risk: Moderate
Why: usually manageable, but concentration and pH shifts matter
Formulation caution: distinguish between precipitation and true crystal growth.
61. Calcium salts in fortified flavored beverages
Use: nutrition-fortified systems
Risk: High
Why: many calcium salts have limited solubility and may seed crystallization or sediment
Formulation caution: not always flavor ingredients themselves, but major contributors to perceived flavor instability.
J. Essential Oil Components with Cold-Solubility Problems
These are often not “classic crystal formers” at room temperature, but they can solidify, haze, or deposit crystals in cold storage, especially in high purity form or when the solvent system shifts.
62. Anethole
Use: anise, fennel, licorice
Risk: High
Why: famous for crystallizing or turning cloudy when diluted or cooled
Formulation caution: one of the most important cold-stability risks in anise systems.
63. Estragole
Use: tarragon, anise herbal
Risk: Moderate
Why: less severe than anethole in many cases, but still sensitive
Formulation caution: cold testing required.
64. D-Carvone
Use: spearmint, caraway
Risk: Low to moderate
Why: often liquid, but purity and temperature affect behavior
Formulation caution: more often haze than strong crystal formation.
65. Pulegone
Use: minty-herbal
Risk: Low to moderate
Why: not usually a major crystal-former
Formulation caution: other concerns often dominate.
66. Citral
Use: lemon, citrus peel
Risk: Low
Why: usually liquid; oxidation is the larger issue
Formulation caution: if solids appear, oxidation byproducts or other components may be involved rather than citral itself.
67. Limonene
Use: citrus
Risk: Low
Why: liquid hydrocarbon terpene
Formulation caution: oxidation and ring formation are bigger issues than crystallization.
K. Encapsulation Carriers and Matrix Materials That May Crystallize
These are not always flavor compounds, but they strongly affect flavor stability.
68. Triacetin
Use: solvent, carrier
Risk: Low
Why: generally stable liquid
Formulation caution: low direct crystallization risk.
69. Propylene glycol
Use: solvent
Risk: Low
Why: remains liquid and helps suppress crystallization of many actives
Formulation caution: useful risk-reduction tool.
70. Glycerol
Use: humectant, solvent
Risk: Low
Why: usually resists classic crystal formation in practical flavor handling
Formulation caution: high water interactions matter more.
71. PEG-type carriers
Use: solvent/carrier
Risk: Moderate
Why: some grades can solidify or crystallize depending on molecular weight
Formulation caution: carrier behavior can unexpectedly trigger flavor drop-out.
72. Starch hydrolysates / maltodextrins
Use: encapsulation matrix
Risk: Low to moderate
Why: often more amorphous than crystalline
Formulation caution: their main issue is glass transition and caking, but some crystallization-related changes can occur indirectly.
Highest-risk compounds for routine flavor work
If a flavorist wants a short watchlist, these are among the most important:
- Menthol
- Vanillin
- Ethyl vanillin
- Maltol
- Ethyl maltol
- Anethole
- Citric acid
- Malic acid
- Tartaric acid
- Fumaric acid
- Sucrose
- Dextrose
- Erythritol
- Mannitol
- Sorbitol
- MSG
- Thymol
- Camphor
- Borneol
- Piperonal
Practical formulation lessons from the library
1. Solids are not always a problem
Some materials are normally sold as crystals or powders. The problem begins when they must remain fully dissolved in the final flavor or food system.
2. Water is often the trigger
A compound may be fully soluble in ethanol or propylene glycol, then crystallize as soon as the flavor is added to a more aqueous beverage.
3. Cold stability matters
Many flavors look perfect at room temperature and fail in the refrigerator or during winter shipping.
4. Purity can increase crystallization
Highly pure compounds often crystallize more easily because impurities no longer disrupt crystal packing.
5. Complex mixtures may help
Sometimes a blend is more stable than a pure material because mixed molecules interfere with crystal growth.
A simple rule-of-thumb risk model
A compound is more likely to crystallize when it has most of these traits:
- solid at room temperature
- high melting point
- aromatic rigidity or symmetry
- hydrogen-bonding groups such as –OH or –COOH
- poor water solubility
- high concentration in concentrate
- exposure to cold temperature
- limited co-solvent support
Bench checklist for flavorists
When working with any moderate- to very-high-risk material, test:
- room temperature clarity
- 4°C / 39°F cold stability
- freeze-thaw behavior
- dilution into final beverage or food matrix
- 1-week and 4-week storage
- microscope check of haze or sediment
- redissolution after warming
- effect of water activity in powders
Summary
A crystallization risk library is useful because flavor instability is often not caused by “chemical spoilage” first. Very often, it begins as a physical stability problem:
- a sweetener crystallizes
- an acid falls out
- a mint solidifies
- a vanilla note seeds crystals
- a clear beverage turns hazy
- a powder cakes and hardens
The compounds with the highest practical risk are usually:
- phenolic sweet solids like vanillin and maltol
- cooling materials like menthol
- organic acids
- polyols and sugars
- certain herbal/aromatic solids like thymol and anethole
Understanding which materials sit in these risk zones helps flavorists choose:
- the right solvent system
- the right loading level
- the right storage conditions
- the right matrix for long shelf life
###