Saponification in Flavor Systems: Mechanism, Stability Determinants, and Practical Control of Ester Hydrolysis in Food and Beverage Formulation
The Society of Flavor Chemists requires flavorists to fully understand approximately two dozen reactions and processes that can occur in flavor systems. Flavorists must be able to control these reactions or physical processes to enhance flavor or improve its stability and shelf life. Saponification is one of the physical processes included among these two dozen reactions and processes.
DEFINITION OF SAPONIFICATION
Saponification in Flavor Chemistry: Definition, Reaction Fundamentals, and Sensory Impact of Ester Hydrolysis
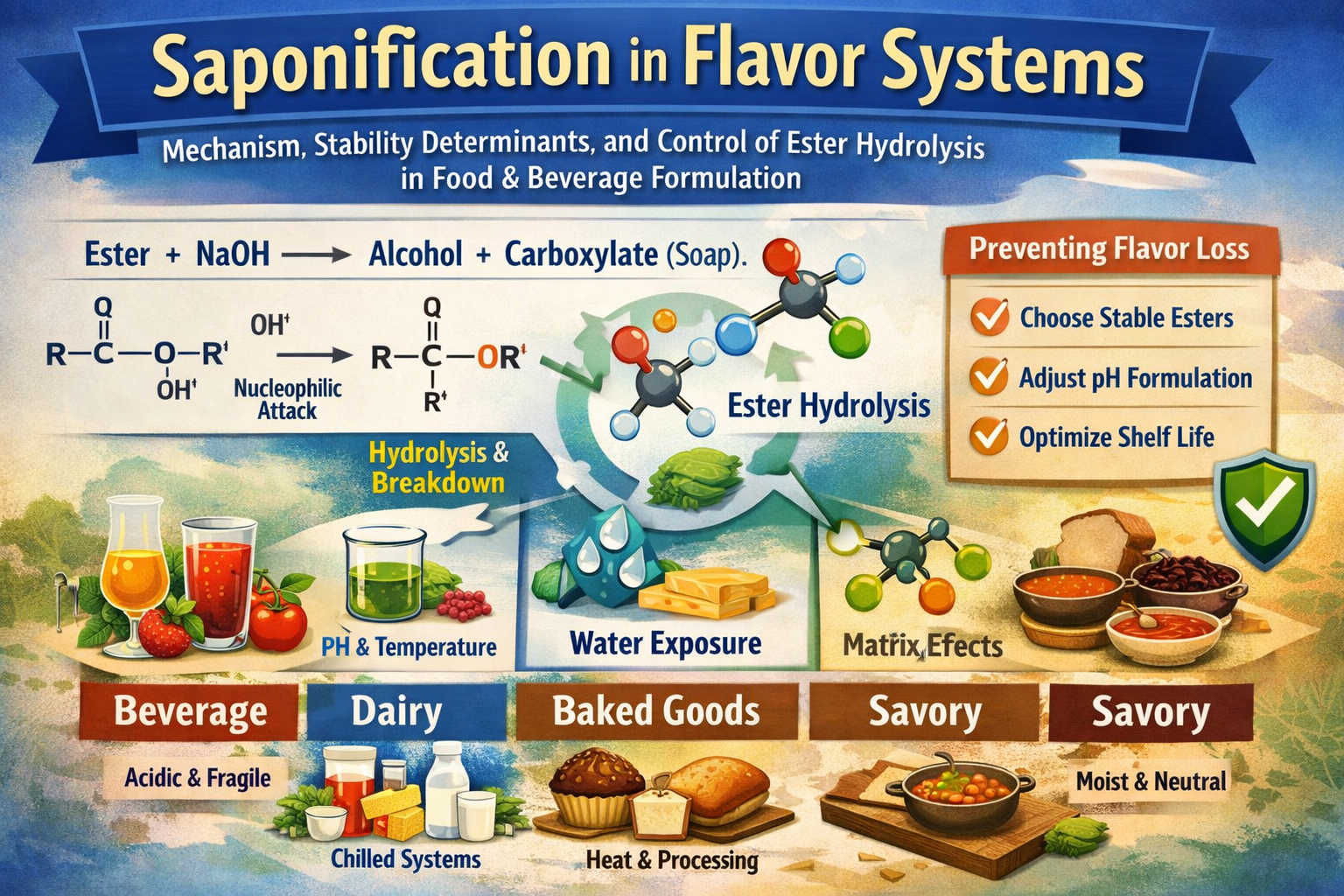
Saponification is a fundamental reaction in organic chemistry and highly relevant to flavor science, especially when working with esters.
🔬 Definition of Saponification
Saponification is the base-catalyzed hydrolysis of an ester, resulting in the formation of:
- an alcohol, and
- a carboxylate salt (commonly called “soap” when derived from fats)
⚗️ Core Reaction Concept
RCOOR' + NaOH \rightarrow RCOO^- Na^+ + R'OH
Where:
- RCOOR′ = ester
- NaOH (or KOH) = strong base
- RCOO⁻ Na⁺ = carboxylate salt (soap-like material)
- R′OH = alcohol
🧪 Simple Explanation (Beginner-Friendly)
Think of an ester as a molecule made by joining:
- an acid part (fatty acid or organic acid), and
- an alcohol part
During saponification:
- A strong base (like sodium hydroxide) attacks the ester bond
- The bond breaks apart
- The molecule splits into:
- a water-soluble salt (soap-like)
- an alcohol
👉 In simple terms:
Saponification = breaking an ester into soap + alcohol using a base
🥼 Classic Example (Fats → Soap)
Natural fats (triglycerides) undergo saponification:
- Fats/oils (esters of glycerol + fatty acids)
- NaOH
→ Glycerol + fatty acid salts (soap)
- NaOH
This is how traditional soap is made.
🍓 Relevance to Flavor Chemistry
Many flavor compounds are esters (e.g., fruity notes). During saponification:
- Fruity esters → destroyed
- Converted into:
- alcohols (often weaker aroma)
- acid salts (usually non-volatile, no aroma)
👉 Result:
- Loss of top notes
- Flavor becomes flat, dull, or soapy
⚠️ Why Flavorists Care
Saponification can occur when:
- pH is alkaline (high pH systems)
- Products contain bases (NaOH, KOH, carbonates)
- During processing or storage
Impact:
- Reduced shelf life
- Off-notes (soapy, fatty)
- Loss of key fruity/floral characteristics
🧾 Summary
- Saponification = base-driven ester breakdown
- Produces alcohol + carboxylate salt
- Critical in:
- soap production
- flavor degradation in alkaline systems
- In flavors: destroys esters → weakens aroma quality
Log in to read more